Kallisio Stentra™ Named “Best New Radiology Technology Solution” in 2026 MedTech Breakthrough Awards Program
Kallisio Stentra™ Named “Best New Radiology Technology Solution” in 2026 MedTech Breakthrough Awards Program
Landmark 10th Annual Awards Program Recognizes the Most Innovative Digital Health and Medical Technology Solutions Worldwide
BALTIMORE--(BUSINESS WIRE)--Kallisio, a company focused on advancing precision oncology through patient-specific, technology-enabled solutions, today announced that Stentra™ has received “Best New Radiology Technology Solution” in the 10th annual MedTech Breakthrough Awards program conducted by MedTech Breakthrough, an independent market intelligence organization that recognizes the most innovative companies, technologies and products in the global digital health and medical technology market.
"Kallisio redefines how precision oncology solutions are designed, governed, and scaled. Radiation should treat cancer without the risk of compromising healthy tissue..."
Share
“Kallisio redefines how precision oncology solutions are designed, governed, and scaled. Radiation should treat cancer without the risk of compromising healthy tissue, but inconsistent immobilization can lead to patient risk. Generic or handcrafted oral appliances are variable in quality, contributing to toxicities, treatment interruptions, and operational inefficiencies,” said Steve Johansson, managing director, MedTech Breakthrough. “Kallisio’s Stentra transforms variability into precision. Combining leadership, evidence, and economic relevance, Stentra is not just digital health; it is digitally enabled medical technology with proven real-world impact.”
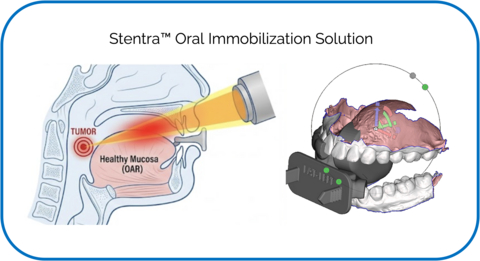
FDA-cleared and CE-marked, Kallisio’s governance-ready Stentra solution combines optical scanning, automated design, and rapid additive manufacturing to deliver millimeter-accurate, 3D-printed devices built specifically for head and neck cancer patient’s oral anatomy and radiation treatment plan. Stentra effectively immobilizes the oral cavity and displaces critical structures such as the tongue. Through consistent tissue displacement and reproducible positioning, the solution drives enhanced radiation dose deliver precision and improved protection of organs-at-risk (OARs). Stentra devices are delivered within 72 hours, supported by ISO 13485-aligned quality systems, and CPT-reimbursable
“Behind every personalized solution is a measurable reduction in patient pain, treatment delays, and clinical workflow complexity, all backed by real-world evidence. Early Stentra data shows up to seven minutes saved per treatment fraction, four hours of LINAC capacity recovered per patient course, and significant decreases in simulation re-scans and unplanned interventions for side effects,” said Rajan Patel, CEO of Kallisio. “We’re honored to be recognized by MedTech Breakthrough, and proud to serve as a strategic partner to hospitals and clinics seeking anatomy-driven solutions that improve outcomes while easing the burden on care teams.”
Honoring the innovations reshaping how care is delivered worldwide, the MedTech Breakthrough Awards program has spent a decade recognizing the companies driving meaningful progress and improving patient care across the global health and medical technology industry.
Contacts
Kallisio Contact
Nicole E. Martin | nicole@kallisio.com | +1.415.860.9194