New Non-Hazardous Viral Inactivation Medium Supports Safe Collection and Transport of SARS-CoV-2
New Non-Hazardous Viral Inactivation Medium Supports Safe Collection and Transport of SARS-CoV-2
Breakthrough formulation helps reduce the risk of exposure using a non-hazardous formulation and leakproof format during transport and processing
BASINGSTOKE, England--(BUSINESS WIRE)--A new viral transport medium that enables collection and rapid inactivation of the SARS-CoV-2 virus, and stabilizes viral RNA at ambient temperature for transportation and use in in vitro diagnostic testing procedures, is now available in Europe.
Thermo Fisher Scientific developed the InhibiSURE Viral Inactivation Medium formula to be non-hazardous. Other inactivation media typically rely on a formulation that is potentially hazardous to inactivate viruses. Many contain a guanidine-based chemical that, if mixed with bleach or strong acids, could release toxic cyanide gas. The use of inactivation medium also removes the need for further inactivation steps, increasing laboratory throughput.
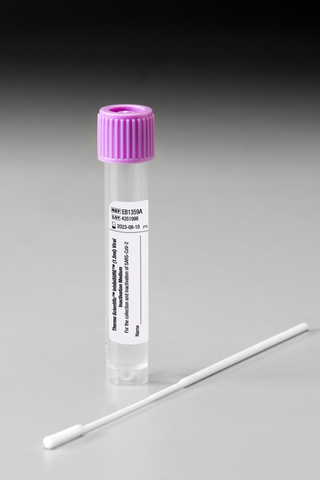
This single-use device, for professional use only, can be used for sampling patients that are suspected of being infected with SARS-CoV-2 or sampling individuals for population screening for SARS-CoV-2.
“As testing demands continue to change dynamically throughout this pandemic, we remain committed to keeping laboratorians safe and protected during the testing process,” said Duncan Porter, general manager, sample collection and transport for Thermo Fisher’s microbiology business. The InhibiSURE Viral Inactivation Medium enables us to support a safe and efficient sample collection and transport process by rapidly inactivating the SARS-CoV-2 virus using a non-hazardous formulation. To broaden the scope of those capabilities, we’ve prioritized the expansion of the range of viruses, as well as the ability to do at-home collection as future developments.”
InhibiSURE Viral Inactivation Medium:
- Helps keep staff safe, inactivating SARS-CoV-2 within 30 minutes of sampling with a non-hazardous formula that does not contain any products that may react with bleach
- Provides a convenient, leakproof, format that can be used with unprocessed nasal swabs, nasopharyngeal swabs or throat swabs on automation preparation racks and decappers
- Enhances simplicity by removing the need for de-activation steps, and is suitable for use with bead and column-based PCR systems
InhibiSURE Viral Inactivation Medium is now commercially available from Thermo Fisher Scientific in Europe, with plans for launch in other regions pending relevant regulatory approvals. For more information, visit thermofisher.com/viraltesting.
Product is CE marked but not 510(k)-cleared and not available for sale in the U.S. Availability of product in each country depends on local regulatory marketing authorization status.
About Thermo Fisher Scientific
Thermo Fisher Scientific Inc. is the world leader in serving science, with annual revenue of approximately $40 billion. Our Mission is to enable our customers to make the world healthier, cleaner and safer. Whether our customers are accelerating life sciences research, solving complex analytical challenges, increasing productivity in their laboratories, improving patient health through diagnostics or the development and manufacture of life-changing therapies, we are here to support them. Our global team delivers an unrivaled combination of innovative technologies, purchasing convenience and pharmaceutical services through our industry-leading brands, including Thermo Scientific, Applied Biosystems, Invitrogen, Fisher Scientific, Unity Lab Services, Patheon and PPD. For more information, please visit www.thermofisher.com.
Contacts
Carlene Graham
Global Marketing Communications Manager
Microbiology
Thermo Fisher Scientific
Phone +1 216.704.3479
carlene.graham@thermofisher.com