Viz.ai Debuts Viz ANX, the First AI-powered Cerebral Aneurysm Detection Solution at SVIN 2021
Viz.ai Debuts Viz ANX, the First AI-powered Cerebral Aneurysm Detection Solution at SVIN 2021
Company also announces Viz SDH for research in subdural hematoma
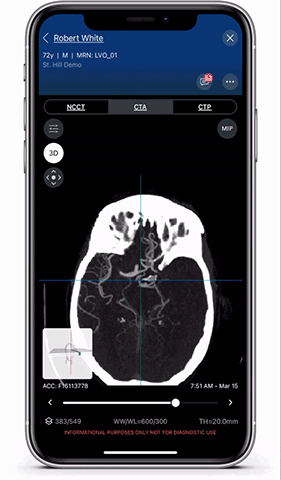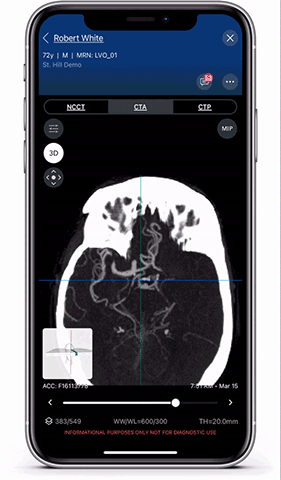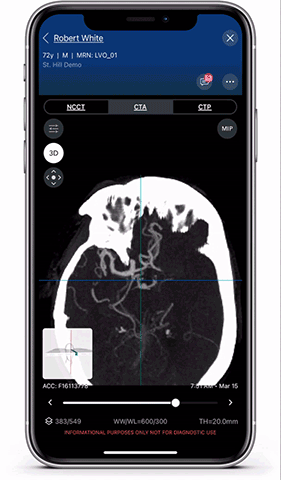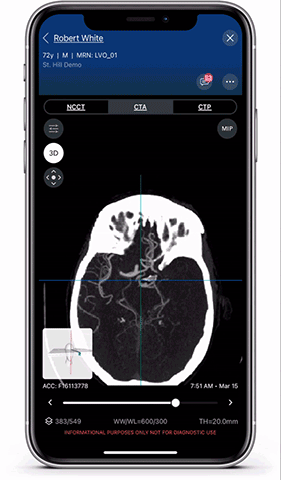
Viz.ai is the leader in AI-powered care coordination and has accelerated the time-to-notification of the treatment team by 73 percent and time-to-treatment by percent. The Viz Platform is now utilized in over 850 hospitals across the U.S. and Europe and touches almost two patients every minute. Shown here is a Viz LVO module viewer. High resolution full 3D viewing enables dynamic visualization of the anatomy for faster decision making and improved care coordination.
SAN FRANCISCO & PHOENIX--(BUSINESS WIRE)--Today at the Society of Vascular and Interventional Neurology (SVIN) 2021 Annual Meeting in Phoenix, Viz.ai, the world leader in AI-powered intelligent care coordination, introduced Viz ANX, the first and only AI-powered suspected cerebral aneurysm detection and care coordination platform. Viz ANX is designed to help ensure every patient who presents with a cerebral aneurysm is quickly identified and receives the correct follow-up, helping prevent a devastating rupture or hemorrhage. The company also introduced Viz SDH, a research-only algorithm supporting the EMBOLISE trial that detects suspected subdural hematoma (SDH) to alert clinicians to potentially eligible study candidates.
Cerebral aneurysm is challenging to detect and treat because current care pathways are complex and rely on manual identification and triage. If left untreated, an aneurysm can burst or rupture, spilling blood into the surrounding tissue, which is an emergent, life-threatening condition. Approximately 30,000 people suffer a rupture in the U.S. every year.
Viz ANX uses artificial intelligence to detect and alert the appropriate care providers of newly detected aneurysms on CTAs. The 510k-pending Viz ANX algorithm will be the first AI software to automatically detect suspected aneurysms. The solution ensures that these aneurysms are captured in an easily accessible and reviewable list and enables one-click referrals to a clinic for follow up, saving valuable time and resources for physicians and helping ensure patients receive proper treatment.
Viz SDH is a research only algorithm that is supporting the EMBOLISE trial. The purpose of the study is to evaluate the safety and efficacy of embolization of the middle meningeal artery (MMA) using the Onyx™ Liquid Embolic System (LES) (made by Medtronic) for treatment of symptomatic subacute or chronic subdural hematoma. The SDH algorithm, which includes volumetric segmentation of and measurement of the maximum thickness of the bleed, detects suspected SDH to alert clinicians to potentially eligible study candidates.
“We know that the Viz platform is widely successful at leveraging AI to help improve workflows and improve patient outcomes in stroke,” said Dr. Ameer Hassan, head of neuroscience and Valley Baptist in Harlingen, TX and president elect of SVIN. “But stroke is just a proof point for how AI can transform patient care and clinical research. By applying the same technology to indications such as aneurysm, I believe we will see further areas of healthcare transformed for better outcomes.”
Viz.ai is the leader in AI-powered care coordination and has accelerated the time-to-notification of the treatment team by 73 percent and time-to-treatment by percent.1 The Viz Platform is now utilized in over 850 hospitals across the U.S. and Europe and touches almost two patients every minute. With the launch of Viz ANX and the support of the EMBOLISE trial, Viz.ai is now leveraging its AI-powered care coordination platform to pioneer aneurysm care and clinical research.
“The introduction of Viz ANX and Viz SDH are part of our ongoing commitment to improve patient outcomes across healthcare,” said Dr. Chris Mansi, co-founder and CEO of Viz.ai. “The value of AI-powered care coordination can help improve workflows, expedite treatment, and ultimately, save lives across so many emergent and less-time sensitive – but equally important – disciplines.”
Viz.ai will showcase the new Viz ANX and Viz SDH, along with the rest of the Viz Intelligent Care Coordination Platform, at SVIN 2021. The company also will host a symposium at SVIN, entitled, “The impact of artificial intelligence on vascular interventional neurology. Today into the future,” featuring Dr. Hassan and Dr. Raul Noguiera, director of neuroendovascular service and neurocritical care service, Marcus Stroke & Neuroscience Center, Grady Memorial Hospital in Atlanta.
About Viz.ai, Inc.
Viz.ai is the leader in AI-driven intelligent care coordination. Viz.ai’s mission is to fundamentally improve how healthcare is delivered in the world, through intelligent software that promises to reduce time to treatment, improve access to care, and increase the speed of diffusion of medical innovation. Viz.ai’s flagship product, Viz LVO, leverages advanced deep learning to communicate time-sensitive information about stroke patients straight to a specialist who can intervene and treat.
In February 2018, the U.S. Food and Drug Administration (FDA) granted a De Novo clearance for Viz LVO, the first-ever computer-aided triage and notification software. Viz.ai announced its second FDA clearance for Viz CTP through the 510(k) pathway, offering healthcare providers an important tool for automated cerebral perfusion image analysis. A third FDA clearance was granted in 2020 for Viz ICH, which uses AI to automatically detect suspected intracranial hemorrhage on CT imaging. Also in 2021, the FDA cleared the Viz PE and Viz Aortic modules, making the Viz Platform multi-disciplinary.
Viz.ai is located in San Francisco and Tel Aviv and backed by leading Silicon Valley investors, including Kleiner Perkins, Google Ventures, Greenoaks, CRV, Threshold Ventures, Innovation Endeavors, Susa Ventures, Scale Venture Partners and Insight Partners. The company was named to the Forbes 2021 Next Billion-Dollar Startups list of the 25 fastest-growing venture-backed startups in 2021 and has been included in the Forbes AI 50 list for the three consecutive years.
1 Elijovich L, DornbosIII D, Nickele C, et al. J NeuroIntervent Surg Epub ahead of print. doi:10.1136/ neurintsurg-2021-017714
Contacts
Joe Duraes
Pazanga Health Communications
jduraes@pazangahealth.com
917-687-6419