MOVIVA® launch spotlight at ESGE Days 2026
MOVIVA® launch spotlight at ESGE Days 2026
“Turn down the food noise”
TÜBINGEN, Germany--(BUSINESS WIRE)--During its 175th anniversary year, Erbe Elektromedizin GmbH highlights its continued commitment to healthcare innovation with the launch of MOVIVA®, a minimally invasive solution for gastric mucosal ablation (GMA) in endoscopic obesity therapy. MOVIVA® will be the central focus of Erbe’s activities at ESGE Days in Milan, 14–16 May 2026. Physicians can explore the technology in the hands-on area and attend a dedicated symposium.
“Obesity continues to rise worldwide, and many patients still face a gap between lifestyle interventions, pharmacotherapy, and bariatric surgery. Endoscopic approaches are emerging as additional treatment options. With MOVIVA®, we aim to support physicians in bringing this innovative treatment option into clinical practice and expanding access to minimally invasive obesity therapies.” Marcus Felstead, Chief Commercial Officer of Erbe.
Targeting hunger at its source
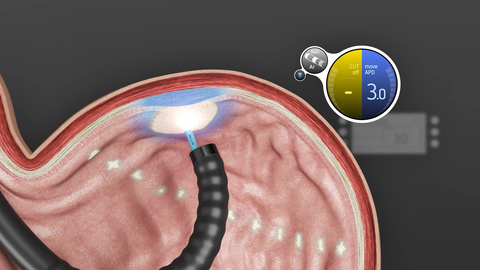
For many people living with obesity, persistent hunger signals and cravings make long-term weight management difficult. The upper part of the stomach - the fundus - is one of the main sites where ghrelin is produced, a hormone that triggers hunger. Gastric mucosal ablation targets this area, reduces production of ghrelin and therefore patients experience less cravings.
MOVIVA® is an advanced hybrid instrument designed for GMA. It combines two key functions in one product. Injection of a submucosal fluid cushion that protects deeper tissue layers from thermal injury and superficial thermal ablation of the mucosa using argon plasma coagulation (APC). MOVIVA® is powered by the moveAPD mode, ensuring broad and consistent ablation.
Early clinical experience with GMA
Clinical evidence indicates that fundal GMA using MOVIVA induces weight loss. GMA alone achieves 7.7% total body weight loss (TBWL) after 6 months in early studies.
GMA may be used either on its own or in combination with endoscopic sleeve gastroplasty (ESG). When combined, early data indicate further improvements, with up to 24% TBWL reported after 12 months – approaching outcomes seen with surgical sleeve gastrectomy, while remaining far less invasive. To date, more than 150 patients have been treated with MOVIVA®, adding to the growing clinical experience with this approach.
Symposium at ESGE Days
At ESGE Days 2026, Erbe will host a symposium with Prof. Aayed Alqahtani, Saudi Arabia, Prof. Ivo Boskoski, Prof. Roberta Maselli, both Italy, and Prof. Bu´Hussain Hayee, UK, titled: “A Novel Endoscopic Therapy for Obesity: Early Clinical Experience with Gastric Mucosal Ablation (GMA) in the fundus.”
The session will provide insights into the early clinical experience with GMA and create a platform for discussing the potential role of this emerging endoscopic therapy in obesity treatment. To stay up to date physicians can also visit the MOVIVA® website [MOVIVA® Turn down the food noise].
MOVIVA® is currently available in CE and CE-related markets.
About Erbe Elektromedizin
In 2026 the Erbe Elektromedizin GmbH celebrates its 175th anniversary under the motto “Progress with Purpose.” Since 1851 Erbe Elektromedizin GmbH develops, manufactures, and distributes surgical instruments and devices worldwide, while providing services for professional use of these products in a diverse range of medical disciplines. Physicians, clinical teams and patients around the world rely on medical technology from Erbe. The instruments and devices find use in almost all specialist areas. They are based on electrosurgery combined with other Erbe technologies. Hybrid solutions enable new, innovative applications in medicine.
Fields of activity
- Electrosurgery
- Endoscopy
- Imaging
- Vessel sealing
- Plasmasurgery
- Cryotechnology
- Hydrosurgery
An international network
- 20 international sales and service units
- 5 production sites
- Erbe is active in 110 international markets
The Erbe workforce
- 2,200 employees worldwide
- Some 1,400 of them in Germany
Contacts
Erbe Elektromedizin GmbH
Waldhoernlestr. 17
72072 Tuebingen
www.erbegroup.com
info@erbegroup.com
Editorial contact
Lisa Viergutz
Phone +49 7071 755-2731
lisa.viergutz@erbegroup.com