New IBS Research Presented at Digestive Disease Week 2026 Validates mBIOTA Elemental as an Effective Therapeutic for Irritable Bowel Syndrome
New IBS Research Presented at Digestive Disease Week 2026 Validates mBIOTA Elemental as an Effective Therapeutic for Irritable Bowel Syndrome
Oral and poster presentations at DDW validate the effectiveness of the mBIOTA Elemental Diet for IBS and other gastrointestinal dysfunctions
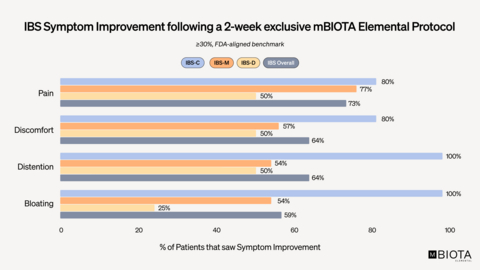
Clinical trial participants across all three subtypes of IBS (constipation, mixed and diarrhea) experienced symptom improvement in abdominal pain, discomfort, distention and bloating after two-weeks of exclusively using mBIOTA Elemental™ Diet. As evaluated using FDA-aligned benchmarks and guidelines, 73% of patients experienced at least a 30% reduction in their daily abdominal pain compared to their baseline.
SANTA MONICA, Calif.--(BUSINESS WIRE)--mBIOTA Labs, an emerging medical food company revolutionizing medical nutrition for the management of gastrointestinal (GI) conditions, announced today that two abstracts featuring mBIOTA Elemental™ Diet are being presented at Digestive Disease Week 2026 in Chicago, Illinois. These abstracts demonstrate the clear effectiveness of mBIOTA Elemental as a therapeutic tool for patients living with Irritable Bowel Syndrome (IBS) and explore the mechanism by which the mBIOTA elemental diet modulates the microbiome in overgrowth conditions.
Notably, 50% of patients had more than or equal to 50% reduction in pain – significantly exceeding the FDA benchmark for improvement of 30%, strongly indicating value in further investigation in a larger-scale clinical trial.
Share
A poster presentation at the event, titled “Abdominal Pain Response to a Two-Week Exclusive Palatable Elemental Diet in Irritable Bowel Syndrome: Post Hoc Analysis of a Prospective Trial,” highlighted mBIOTA Elemental as a highly impactful therapeutic tool for managing IBS. Responses in abdominal pain, discomfort, distention and bloating were measured in patients with all three subtypes of IBS (constipation, mixed and diarrhea). As evaluated using FDA-aligned benchmarks and guidelines, 73% of patients experienced at least a 30% reduction in their daily abdominal pain compared to their baseline. Notably, 50% of patients had more than or equal to 50% reduction in pain – significantly exceeding the FDA benchmark for improvement of 30%, strongly indicating value in further investigation in a larger-scale clinical trial.
The findings underscore mBIOTA Elemental’s success across all three subtypes of IBS, adding to the depth of clinical insight for the use of a palatable elemental diet for IBS. Other notable findings include:
- 82% of patients achieved a statistically significant improvement in at least one symptom (pain, distention, discomfort, bloating)
- 73% of IBS patients had a ≥30% reduction (IBS-C 80%, IBS-M 77%, IBS-D 50%) in pain
- 100% of IBS-C patients achieved clinically meaningful relief (≥30%) from bloating and distention
- 77% of IBS-M patients experienced significant (≥30%) pain improvement
“We are extremely proud to share the IBS patient symptom reduction findings at DDW 2026 as yet another rare achievement across GI clinical trials, following our groundbreaking success in setting a new standard for patient compliance rates,” said Nicola Wodlinger, CEO of mBIOTA Labs. “IBS is a prevalent condition, yet can still be difficult to effectively manage with its wide-ranging symptoms. This new clinical evidence validates mBIOTA Elemental Diet as an effective therapeutic tool for IBS. mBIOTA Labs is committed to improving GI patients’ quality of life through scientifically-backed solutions and expanding clinical insight supporting the power of medical nutrition for those with IBS and beyond.”
Additionally, an oral presentation titled, “Effect of Exclusive Palatable Elemental Diet on Systemic Inflammation and Gut Microbiome in Microbial Overgrowth Syndromes: Ad Hoc Analysis of a Prospective Trial,” explores changes in stool microbiome and inflammatory markers of patients following 2 weeks of the mBIOTA Elemental Diet. The findings affirm that microbiome modulation is a key underlying mechanism driving symptom relief in patients with small intestinal bacterial overgrowth (SIBO) and intestinal methanogen overgrowth (IMO). Further, populations of microbial species associated with such dysbiosis, including Methanobrevobacter smithii and Candida albicans, remained decreased after reintroduction of a regular diet.
The exceptionally strong IBS efficacy findings presented at DDW 2026 signal promising potential for future clinical trial research that can meaningfully redefine therapeutic outcome expectations for IBS patients. mBIOTA Labs’ palatable elemental diet offers patients an approachable and clinically reliable option to manage GI conditions like IBS, SIBO, and IMO. For more information on mBIOTA Labs, mBIOTA Elemental, and clinical findings validating its microbiome modulation and more, visit www.mBIOTA.com.
About mBIOTA Labs
mBIOTA Labs empowers individuals with evidence-based nutrition solutions that enhance quality of life. Trusted by the medical community, mBIOTA Labs harnesses proprietary Amino Taste Modification Technology (ATMT) to create science-backed medical foods designed to support individuals with a wide range of digestive issues. mBIOTA's award-winning, clinically proven, palatable elemental diet, mBIOTA Elemental™, revolutionizes medical nutrition for the dietary management of GI dysfunction, including small intestinal bacterial overgrowth (SIBO), intestinal methanogen overgrowth (IMO), irritable bowel syndrome (IBS), inflammatory bowel diseases (IBD) such as Crohn’s disease and colitis, eosinophilic esophagitis (EoE), and other conditions. mBIOTA is actively investigating and researching expanded applications of the elemental diet to aid and provide solutions to more patients.
Learn more about mBIOTA Labs and published research supporting mBIOTA Elemental Diet by visiting www.mBIOTA.com.
Contacts
Media Contact:
Sarah Browning
Public Relations for mBIOTA Labs
mbiota@matternow.com