Masimo Announces Preliminary Second Quarter 2023 Revenue Results
Masimo Announces Preliminary Second Quarter 2023 Revenue Results
Management to announce and discuss complete second quarter 2023 financial results on Tuesday, August 8, 2023.
IRVINE, Calif.--(BUSINESS WIRE)--Masimo Corporation (Nasdaq: MASI) today announced preliminary revenue results for the second quarter ended July 1, 2023.
Preliminary Second Quarter 2023 Revenue Results
Masimo expects its consolidated revenue for the second quarter 2023 to range from $453 million to $457 million, with healthcare revenue expected to range from $280 million to $282 million and non-healthcare revenue expected to range from $173 million to $175 million.
Though the healthcare business made significant market share gains through new contracting in the second quarter, healthcare revenue for the second quarter 2023 was lower than expected due to multiple factors, including the following:
- Large orders that were anticipated for the second quarter were delayed to the second half of the year.
-
Single-patient use sensor sales were down due to:
- Lower than expected U.S. hospital inpatient census, which drives usage of single-patient use sensors; and
- Elevated sensor inventory levels at some customers due to discounting in prior quarters, which was discontinued during the second quarter, and the abnormally early end of the flu season, which faded quickly in the first quarter this year.
- Conversions of new customers who have contracted to switch to Masimo were less than expected due to labor shortages in hospitals, and our OEM partners not being able to provide the patient monitoring equipment needed to complete the installations in a timely manner.
- Continued increased hospital labor costs have strained hospital budgets, lowering demand for capital equipment in the second quarter.
Non-healthcare revenue for the second quarter 2023 fell below expectations as the decline in demand previously seen in lower-end consumer audio categories extended into the premium and luxury audio categories and across more geographies.
Preliminary Outlook
With second quarter revenues below expectations, the Company is taking actions to reduce costs in the second half of the year. Management will share more details on these actions when the company announces complete second quarter 2023 financial results on Tuesday, August 8, 2023.
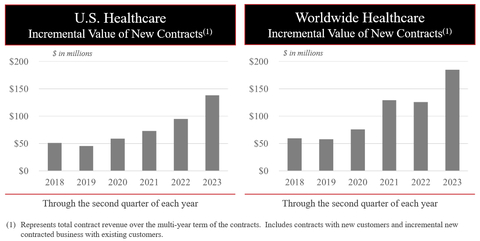
Despite the second quarter revenue shortfall, the fundamentals of both businesses remain strong. Notably, on the healthcare side, new hospital customers continue to switch to Masimo technology faster than ever, increasing Masimo’s share of the hospital market. Masimo drove record contracting in the first half of the year, both in the U.S. and worldwide.
Masimo’s Unrecognized Contract Revenue (as defined in Masimo’s Annual Report on Form 10-K filed March 1, 2023) is expected to range from $1.37 billion to $1.38 billion at the end of the second quarter, representing growth of 11% to 12% in Unrecognized Contract Revenue versus the end of the second quarter of 2022.
On the consumer side, it is unclear when the softness in the premium and luxury categories will improve. However, initial demand for the Stork™ baby monitor has been strong, as major retailers are placing stocking orders for the product. In addition, hearables sales doubled this quarter versus the same quarter last year, helped by the launch of the Denon PerL™ earbuds with Masimo AAT™.
Joe Kiani, Chairman and CEO of Masimo, stated, “While we are disappointed in our revenue results this quarter, our hospital business is strong, as our growth in contracting shows. We do believe sensor utilization and sensor revenue growth rates will return to normal levels. We are also excited about the future for Radius VSM™, Opioid Halo™, Stork™, PerL™, W1™, and our telemonitoring businesses, all of which are in full-scale launch, except for Stork™, which is in limited market release.”
Management plans to discuss Masimo’s complete second quarter 2023 financial results and provide updated fiscal year 2023 financial guidance after the market closes on Tuesday, August 8, 2023. We currently expect to reduce the lower end of full year revenue guidance for the healthcare business to $1.30 billion from $1.45 billion. We are still evaluating the upper end of revenue guidance for the healthcare business, but it could be materially higher than the lower end of the range, as we are still targeting our original guidance. We also expect to reduce annual revenue guidance for the non-healthcare business to $800 million to $850 million from $965 million to $995 million.
The preliminary financial information presented in this press release is based on Masimo’s current expectations and may be adjusted as a result of the completion of customary quarterly review procedures and other processes.
Conference Call:
The conference call to review Masimo’s complete second quarter 2023 results will begin at 1:30 p.m. PT (4:30 p.m. ET) on Tuesday, August 8, 2023, and will be hosted by Joe Kiani, Chairman and Chief Executive Officer, and Micah Young, Executive Vice President and Chief Financial Officer.
To register for the conference call and receive the dial-in number, please use the link below. Upon registering, each participant will be provided with call details and a registrant ID number.
Conference Call Registration Link:
https://conferencingportals.com/event/nUSpRIEm
A replay of the webcast and conference call will be available shortly after the conclusion of the call and will be archived on the Company’s website.
About Masimo
Masimo (Nasdaq: MASI) is a global technology company that develops and produces a wide array of industry-leading monitoring technologies, including innovative measurements, sensors, patient monitors, and automation and connectivity solutions. In addition, Masimo Consumer Audio is home to eight iconic audio brands, including Bowers & Wilkins®, Denon®, Marantz®, and Polk Audio®. Our mission is to improve life, improve patient outcomes; and reduce the cost of care. Masimo SET® Measure-through Motion and Low Perfusion™ pulse oximetry, introduced in 1995, has been shown in over 100 independent and objective studies to outperform other pulse oximetry technologies. Masimo SET® has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates, improve CCHD screening in newborns, and, when used for continuous monitoring with Masimo Patient SafetyNet™ in post-surgical wards, reduce rapid response team activations, ICU transfers, and costs. Masimo SET® is estimated to be used on more than 200 million patients in leading hospitals and other healthcare settings around the world, and is the primary pulse oximetry at 9 of the top 10 hospitals as ranked in the 2022-23 U.S. News and World Report Best Hospitals Honor Roll. In 2005, Masimo introduced rainbow® Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively and intermittently, including total hemoglobin (SpHb®), oxygen content (SpOC™), carboxyhemoglobin (SpCO®), methemoglobin (SpMet®), Pleth Variability Index (Pvi®), RPVi™ (rainbow® Pvi), and Oxygen Reserve Index (Ori™). In 2013, Masimo introduced the Root® Patient Monitoring and Connectivity Platform, built from the ground up to be as flexible and expandable as possible to facilitate the addition of other Masimo and third-party monitoring technologies; key Masimo additions include Next Generation SedLine® Brain Function Monitoring, O3® Regional Oximetry, and ISA™ Capnography with NomoLine® sampling lines. Masimo’s family of continuous and spot-check monitoring Pulse CO-Oximeters® includes devices designed for use in a variety of clinical and non-clinical scenarios, including tetherless, wearable technology, such as Radius-7®, Radius-PPG® and Radius VSM™, portable devices like Rad-67®, fingertip pulse oximeters like MightySat® Rx, and devices available for use both in the hospital and at home, such as Rad-97®. Masimo hospital and home automation and connectivity solutions are centered around Root and the Masimo Hospital Automation™ platform, and include Iris® Gateway, iSirona™, Patient SafetyNet, Replica®, Halo ION, UniView®, UniView :60™, and Masimo SafetyNet™. Masimo’s growing portfolio of health and wellness solutions include Radius T™ and the Masimo W1™ watch, Stork™, Opioid Halo™, Bridge™, and PerL™. Additional information about Masimo and its products may be found at www.masimo.com. Published clinical studies on Masimo products can be found at www.masimo.com/evidence/featured-studies/feature/.
Ori and RPVi have not received FDA 510(k) clearance and are not available for sale in the United States. The use of the trademark Patient SafetyNet is under license from University HealthSystem Consortium.
Forward-Looking Statements
All statements other than statements of historical facts included in this press release that address activities, events or developments that we expect, believe or anticipate will or may occur in the future are forward-looking statements including our expectations regarding our second quarter 2023 consolidated revenue, healthcare revenue and non-healthcare revenue, our expected timing for announcing complete second quarter 2023 financial results and updated fiscal 2023 financial guidance, our preliminary adjustments to our fiscal 2023 healthcare and consumer revenue guidance, our expectations regarding Unrecognized Contract Revenue, our anticipated receipt of large orders and the timing of receipt or any such orders, potential cost reductions, our expectation that sensor utilization and sensor revenue growth rates will return to normal levels and our future opportunities in the pipeline. These forward-looking statements are based on management’s current expectations and beliefs and are subject to uncertainties and factors, all of which are difficult to predict and many of which are beyond our control and could cause actual results to differ materially and adversely from those described in the forward-looking statements. These risks include, but are not limited to, those related to: the completion of customary quarterly financial statement review procedures; our dependence on Masimo SET® and Masimo rainbow SET™ products and technologies for substantially all of our revenue; any failure in protecting our intellectual property exposure to competitors’ assertions of intellectual property claims; the highly competitive nature of the markets in which we sell our products and technologies; any failure to continue developing innovative products and technologies; our ability to successfully integrate Sound United’s brands into our business; our ability to address and expand into new markets; the lack of acceptance of any of our current or future products and technologies; obtaining regulatory approval of our current and future products and technologies; the risk that the implementation of our international realignment will not continue to produce anticipated operational and financial benefits, including a continued lower effective tax rate; the loss of our customers; the failure to retain and recruit senior management; product liability claims exposure; a failure to obtain expected returns from the amount of intangible assets we have recorded; the maintenance of our brand; the amount and type of equity awards that we may grant to employees and service providers in the future; our ongoing litigation and related matters; and other factors discussed in the “Risk Factors” section of our most recent periodic reports filed with the Securities and Exchange Commission (“SEC”), including our most recent Form 10-K and Form 10-Q, all of which you may obtain for free on the SEC’s website at www.sec.gov. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof, even if subsequently made available by us on our website or otherwise. We do not undertake any obligation to update, amend or clarify these forward-looking statements, whether as a result of new information, future events or otherwise, except as may be required under applicable securities laws.
Masimo, SET, Signal Extraction Technology, Improving Patient Outcome and Reducing Cost of Care... by Taking Noninvasive Monitoring to New Sites and Applications, rainbow, SpHb, SpOC, SpCO, SpMet, PVI and ORI are trademarks or registered trademarks of Masimo Corporation.
Contacts
Investor Contact: Eli Kammerman
(949) 297-7077
ekammerman@masimo.com
Media Contact: Evan Lamb
(949) 396-3376
elamb@masimo.com