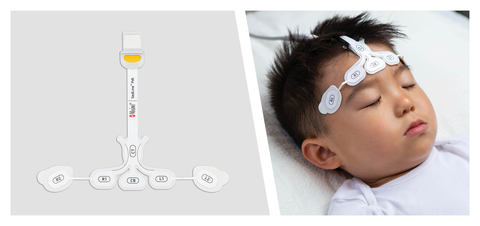
Randomized Controlled Study Finds Masimo SedLine® Brain Function Monitoring Can Help Guide Anesthesia in Children Undergoing Minor Surgery
Randomized Controlled Study Finds Masimo SedLine® Brain Function Monitoring Can Help Guide Anesthesia in Children Undergoing Minor Surgery
Anesthesiologists Used Less Sevoflurane to Maintain Appropriate Anesthesia in Children Monitored with Masimo SedLine
NEUCHATEL, Switzerland--(BUSINESS WIRE)--Masimo (NASDAQ: MASI) today announced the findings of a randomized, controlled trial published in the Journal of Clinical Anesthesia in which Dr. Melody H.Y. Long and colleagues from the KK Women’s and Children’s Hospital in Singapore evaluated the ability of electroencephalogram (EEG)-guided anesthesia, using Masimo SedLine® brain function monitoring, to reduce the amount of the drug sevoflurane needed to maintain anesthesia in pediatric patients undergoing minor surgery.1 They found that use of SedLine to guide anesthesia reduced sevoflurane requirements and led to a reduced incidence of burst suppression, which has previously been reported to be associated with postoperative delirium.2-7
Noting the unique nature of pediatric brains, which are still developing, the importance that standard anesthesia practice places on minimizing the dosage of drugs needed to maintain anesthesia, and the lack of research into the use of new technology like real-time EEG spectrogram monitoring in children, the researchers devised a study that would investigate what impact such technology might have. They enrolled 195 children, aged 1 to 6 years, who were scheduled for minor surgery involving general anesthesia induced and maintained using sevoflurane. The children were randomized into either a Masimo SedLine EEG-guided group (n=100) or a standard care group (n=95). In the SedLine EEG group, anesthesiologists used SedLine to help guide administration of sevoflurane, with the goals of maintaining continuous slow/delta oscillations on the raw EEG and spectrogram, avoiding burst suppression, and maintaining a Patient State Index, or PSi – a propriety, processed EEG parameter developed by Masimo – between 25 and 50. In the standard care group, clinicians were blinded to the EEG data.
As their primary outcome, the researchers looked at the average end-tidal concentration of sevoflurane used during induction and maintenance of anesthesia. They found that in the EEG group, the concentration was lower both during induction (4.80% compared to 5.67% in the control group, p=0.003) and maintenance (2.23% vs. 2.38%, p=0.005). As one of their secondary outcomes, the researchers compared the incidence and duration of intraoperative burst suppression, and found that the EEG group had a lower incidence of burst suppression (3.1% vs. 10.9% in the control group, p=0.0440).
The authors concluded, “This is one of the first randomized control trials in the pediatric population showing that EEG-guided anesthesia care utilizing the spectrogram is feasible, and leads to a modest decrease in intraoperative sevoflurane dosage for induction and maintenance in young children aged 1 to 6 years. EEG guidance allows easy visualization of anesthesia-induced changes on the brain in real time, making it possible to determine which individuals require more (or less) anesthetic to maintain unconsciousness and titrate doses accordingly. This may be particularly important in children between 1 and 2 years old, who appear to require a higher concentration of sevoflurane during surgery, as well as in patients at risk of neurological injury. Our findings highlight the importance of EEG monitoring in complementing the current ASA standard monitors, to provide personalized anesthesia care.”
William C. Wilson, MD, MA, CMO and SVP of Clinical Research and Medical Affairs at Masimo, commented, “We believe the significant reduction in burst suppression noted in the EEG group – less than one-third the amount in the control group – is an important finding. In future studies with larger sample pools, this could demonstrate more profound outcome benefits.”
In the U.S., SedLine is currently indicated for pediatric use without the PSi parameter.
@Masimo | #Masimo
About Masimo
Masimo (NASDAQ: MASI) is a global medical technology company that develops and produces a wide array of industry-leading monitoring technologies, including innovative measurements, sensors, patient monitors, and automation and connectivity solutions. Our mission is to improve patient outcomes and reduce the cost of care. Masimo SET® Measure-through Motion and Low Perfusion™ pulse oximetry, introduced in 1995, has been shown in over 100 independent and objective studies to outperform other pulse oximetry technologies.8 Masimo SET® has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates,9 improve CCHD screening in newborns,10 and, when used for continuous monitoring with Masimo Patient SafetyNet™ in post-surgical wards, reduce rapid response team activations, ICU transfers, and costs.11-14 Masimo SET® is estimated to be used on more than 200 million patients in leading hospitals and other healthcare settings around the world,15 and is the primary pulse oximetry at 9 of the top 10 hospitals as ranked in the 2021-22 U.S. News and World Report Best Hospitals Honor Roll.16 Masimo continues to refine SET® and in 2018, announced that SpO2 accuracy on RD SET® sensors during conditions of motion has been significantly improved, providing clinicians with even greater confidence that the SpO2 values they rely on accurately reflect a patient’s physiological status. In 2005, Masimo introduced rainbow® Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb®), oxygen content (SpOC™), carboxyhemoglobin (SpCO®), methemoglobin (SpMet®), Pleth Variability Index (PVi®), RPVi™ (rainbow® PVi), and Oxygen Reserve Index (ORi™). In 2013, Masimo introduced the Root® Patient Monitoring and Connectivity Platform, built from the ground up to be as flexible and expandable as possible to facilitate the addition of other Masimo and third-party monitoring technologies; key Masimo additions include Next Generation SedLine® Brain Function Monitoring, O3® Regional Oximetry, and ISA™ Capnography with NomoLine® sampling lines. Masimo’s family of continuous and spot-check monitoring Pulse CO-Oximeters® includes devices designed for use in a variety of clinical and non-clinical scenarios, including tetherless, wearable technology, such as Radius-7® and Radius PPG™, portable devices like Rad-67®, fingertip pulse oximeters like MightySat® Rx, and devices available for use both in the hospital and at home, such as Rad-97®. Masimo hospital automation and connectivity solutions are centered around the Masimo Hospital Automation™ platform, and include Iris® Gateway, iSirona™, Patient SafetyNet, Replica®, Halo ION™, UniView®, UniView :60™, and Masimo SafetyNet®. In 2022, Masimo acquired Sound United, a leading developer of premium consumer sound and home integration technologies, whose brands include Bowers & Wilkins®, Denon®, Polk Audio®, Marantz®, Definitive Technology®, Classé®, and Boston Acoustics®. Additional information about Masimo and its products may be found at www.masimo.com. Published clinical studies on Masimo products can be found at www.masimo.com/evidence/featured-studies/feature/.
ORi and RPVi have not received FDA 510(k) clearance and are not available for sale in the United States. The use of the trademark Patient SafetyNet is under license from University HealthSystem Consortium.
References
- Long MHY, Lim EHL, Balanza GA, Allen JC, Purdon PL, and Bong CL. Sevoflurane requirements during electroencephalogram (EEG)-guided vs. standard anesthesia care in children: A randomized controlled trial. J Clin Anesth. 27 Jun 2022. DOI: https://doi.org/10.1016/j.jclinane.2022.110913,
- Tang CJ, Jin Z, Sands LP, Pleasants D, Tabatabai S, Hong Y, Leung JM. ADAPT-2: A Randomized Clinical Trial to Reduce Intraoperative EEG Suppression in Older Surgical Patients Undergoing Major Noncardiac Surgery. Anesth Analg 2020; 131(4):1228-1236.
- Radtke FM, Franck M, Lendner J, Krüger S, Wernecke KD, Spies CD. Monitoring depth of anaesthesia in a randomized trial decreases the rate of postoperative delirium but not postoperative cognitive dysfunction. Br J Anaesthesia. (2013) 110:98–105. doi: 10.1093/bja/aet055.
- MacKenzie KK, Britt-Spells AM, Sands LP, Leung JM. Processed electroencephalogram monitoring and postoperative delirium: a systematic review and meta-analysis. Anesthesiology. (2018)129:417–27. doi: 10.1097/ALN.0000000000002323.
- Sieber FE, Zakriya KJ, Gottschalk A, Blute MR, Lee HB, Rosenberg PB, et al. Sedation depth during spinal anesthesia and the development of postoperative delirium in elderly patients undergoing hip fracture repair. Mayo Clin Proc. (2010) 85:18–26. doi: 10.4065/mcp.2009.0469.
- Whitlock EL, Torres BA, Lin N, Helsten DL, Nadelson MR, Mashour GA, et al. Postoperative delirium in a substudy of cardiothoracic surgical patients in the BAG-RECALL clinical trial. Anesth Analg. (2014) 118:809–17. doi: 10.1213/ANE.000000000000002.
- Xu N, Li L, Wang T, Jiao L, Hua Y, Yao D, Wu J, Ma Y, Tian T, Sun X. Processed Multiparameter Electroencephalogram-Guided General Anesthesia Management Can Reduce Postoperative Delirium Following Carotid Endarterectomy: A Randomized Control Trial. Front Neurol. 12 July 2021. 12:666814. doi: 10.3389/fneur.2021.666814.
- Published clinical studies on pulse oximetry and the benefits of Masimo SET® can be found on our website at http://www.masimo.com. Comparative studies include independent and objective studies which are comprised of abstracts presented at scientific meetings and peer-reviewed journal articles.
- Castillo A et al. Prevention of Retinopathy of Prematurity in Preterm Infants through Changes in Clinical Practice and SpO2 Technology. Acta Paediatr. 2011 Feb;100(2):188-92.
- de-Wahl Granelli A et al. Impact of pulse oximetry screening on the detection of duct dependent congenital heart disease: a Swedish prospective screening study in 39,821 newborns. BMJ. 2009;Jan 8;338.
- Taenzer A et al. Impact of pulse oximetry surveillance on rescue events and intensive care unit transfers: a before-and-after concurrence study. Anesthesiology. 2010:112(2):282-287.
- Taenzer A et al. Postoperative Monitoring – The Dartmouth Experience. Anesthesia Patient Safety Foundation Newsletter. Spring-Summer 2012.
- McGrath S et al. Surveillance Monitoring Management for General Care Units: Strategy, Design, and Implementation. The Joint Commission Journal on Quality and Patient Safety. 2016 Jul;42(7):293-302.
- McGrath S et al. Inpatient Respiratory Arrest Associated With Sedative and Analgesic Medications: Impact of Continuous Monitoring on Patient Mortality and Severe Morbidity. J Patient Saf. 2020 14 Mar. DOI: 10.1097/PTS.0000000000000696.
- Estimate: Masimo data on file.
- http://health.usnews.com/health-care/best-hospitals/articles/best-hospitals-honor-roll-and-overview.
Forward-Looking Statements
This press release includes forward-looking statements as defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, in connection with the Private Securities Litigation Reform Act of 1995. These forward-looking statements include, among others, statements regarding the potential effectiveness of Masimo SedLine®. These forward-looking statements are based on current expectations about future events affecting us and are subject to risks and uncertainties, all of which are difficult to predict and many of which are beyond our control and could cause our actual results to differ materially and adversely from those expressed in our forward-looking statements as a result of various risk factors, including, but not limited to: risks related to our assumptions regarding the repeatability of clinical results; risks related to our belief that Masimo's unique technologies, including Masimo SedLine, contribute to positive clinical outcomes and patient safety; risks that the researchers’ conclusions and findings may be inaccurate; risks related to our belief that Masimo noninvasive medical breakthroughs provide cost-effective solutions and unique advantages; risks related to COVID-19; as well as other factors discussed in the "Risk Factors" section of our most recent reports filed with the Securities and Exchange Commission ("SEC"), which may be obtained for free at the SEC's website at www.sec.gov. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. All forward-looking statements included in this press release are expressly qualified in their entirety by the foregoing cautionary statements. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of today's date. We do not undertake any obligation to update, amend or clarify these statements or the "Risk Factors" contained in our most recent reports filed with the SEC, whether as a result of new information, future events or otherwise, except as may be required under the applicable securities laws.
Contacts
Media Contact:
Masimo
Evan Lamb
949-396-3376
elamb@masimo.com