Annovis Bio Measures Novel Biomarkers in Plasma of Parkinson’s Patients
Annovis Bio Measures Novel Biomarkers in Plasma of Parkinson’s Patients
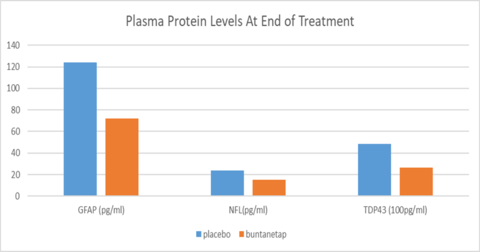
Buntanetap lowers the levels of TDP43, NfL, and GFAP
BERWYN, Pa.--(BUSINESS WIRE)--Recent developments in biomarker research have enabled the performance of measurements of important biomarkers in plasma rather than in CSF (cerebrospinal fluid), making it possible to follow the changes in biomarkers during the course of a neurodegenerative disease while reducing patients’ burden.
TDP-43 (TAR DNA-binding protein 43) was discovered by Lee & Trojanowski at the University of Pennsylvania in 2007, and was suggested to be associated with frontotemporal dementia (FTD, 1),. Shaw’s lab in King’s College London associated TDP43 with amyotrophic lateral sclerosis published in Science 2008 (ALS, 2). More recently a number of papers in high profile journals have associated the protein with other neurodegenerative diseases, such as Alzheimer’s disease (AD, 3,4,5,8,9) and Parkinson’s disease (PD, 6,7). Of special interest, Dr. Ron Peterson from the Mayo Clinic pointed out in his 2018 Neurology review that protein abnormalities beyond amyloid and tau, including TDP-43 and α-synuclein, may contribute to neurodegeneration and cognitive impairment (5).
TDP-43 turns out to be a neurotoxic aggregating protein that has a normal function at normal levels, but when overexpressed it aggregates and becomes toxic, just like Aβ and α-synuclein. It impairs axonal transport, induces inflammation, and kills nerve cells.
Two years ago, Annovis found that buntanetap inhibited expression of TDP43 in vitro through an unbiased proteomics search of proteins whose translation is regulated by buntanetap as published (10) and in table below. Now we were able to see a decrease of TDP43 in plasma of PD patients from our phase 2a study. This was made possible through the new Quanterix Simoa TDP43 assay, which detects both full-length and pathological, truncated forms of the protein. The data revealed that 80mg buntanetap treatment (n=10 patients) indeed reduced the accumulation of TDP43 in patients’ plasma by 71.7% compared to placebo (n=5 patients), with a statistically significant difference between placebo and treated p=0.05. Buntanetap also showed a trend in reducing inflammatory factor GFAP (Glial fibrillary acidic protein) and the axonal damage biomarker NfL (Neurofilament-light chain).
Table shows changes in protein levels of neurotoxic proteins whose mRNAs contain an atypical IRE (iron-responsive-element) and control proteins whose mRNAs contain a canonical IRE (FTL, and FHL1) in their 5′-UTR following buntanetap treatment of SH-SY5Y cells. The column with the ratios shows by how much the protein level goes down after the cells have been treated with buntanetap. This data is from our unbiased proteomics study.
Symbol |
Gene Name |
After/Before Treatment |
HTT |
Huntingtin |
0.479 |
APP |
Amyloid beta protein |
0.728 |
TARDBP |
TAR DNA-binding protein 43 (TDP43) |
0.761 |
SNCA |
Alpha-synuclein |
0.779 |
FTL |
Ferritin light chain |
0.978 |
FTH1 |
Ferritin heavy chain |
0.975 |
These exciting new data support buntanetap’s unique mechanism of action of reducing over-expression of multiple neurotoxic proteins in disease situations, therefore, reducing inflammation and neuronal damage, and preserving the neuronal function.
“We are pleasantly surprised to see a statistically significant drop in TDP43 levels in just 10 patients and to see a strong trend in GFAP and NfL. To our knowledge this is the first time that a drug reduces the levels of TDP43 in humans, specifically here in PD patients,” said Dr. Maccecchini. “These biomarker data not only corroborate the mechanism of actions of buntanetap, but also provide a new way to stratify patients and understand their disease pathology. We will measure the above biomarkers in the plasma of the patients that are presently in our phase 3 PD study to show the effect our drug has on the course of the disease over a six-month period.”
- Cairns NJ, Neumann M, Bigio EH, Holm IE, Troost D, Hatanpaa KJ, Foong C, White CL 3rd, Schneider JA, Kretzschmar HA, Carter D, Taylor-Reinwald L, Paulsmeyer K, Strider J, Gitcho M, Goate AM, Morris JC, Mishra M, Kwong LK, Stieber A, Xu Y, Forman MS, Trojanowski JQ, Lee VM, Mackenzie IR. TDP-43 in familial and sporadic frontotemporal lobar degeneration with ubiquitin inclusions. Am J Pathol. 2007 Jul;171(1):227-40. doi: 10.2353/ajpath.2007.070182. PMID: 17591968; PMCID: PMC1941578. link
- Sreedharan J, Blair IP, Tripathi VB, Hu X, Vance C, Rogelj B, Ackerley S, Durnall JC, Williams KL, Buratti E, Baralle F, de Belleroche J, Mitchell JD, Leigh PN, Al-Chalabi A, Miller CC, Nicholson G, Shaw CE. TDP-43 mutations in familial and sporadic amyotrophic lateral sclerosis. Science. 2008 Mar 21;319(5870):1668-72. doi: 10.1126/science.1154584. Epub 2008 Feb 28. PMID: 18309045; PMCID: PMC7116650. link
- Meneses A, Koga S, O'Leary J, Dickson DW, Bu G, Zhao N. TDP-43 Pathology in Alzheimer's Disease. Mol Neurodegener. 2021 Dec 20;16(1):84. doi: 10.1186/s13024-021-00503-x. PMID: 34930382; PMCID: PMC8691026. link
- Josephs KA, Whitwell JL, Tosakulwong N, Weigand SD, Murray ME, Liesinger AM, Petrucelli L, Senjem ML, Ivnik RJ, Parisi JE, Petersen RC, Dickson DW. TAR DNA-binding protein 43 and pathological subtype of Alzheimer's disease impact clinical features. Ann Neurol. 2015 Nov;78(5):697-709. doi: 10.1002/ana.24493. Epub 2015 Sep 16. PMID: 26224156; PMCID: PMC4623932. link
- Peterson R. How early can we diagnose Alzheimer disease (and is it sufficient)? Neurology® 2018;91:395-402. link
- Yamashita, R., Beck, G., Yonenobu, Y., Inoue, K., Mitsutake, A., Ishiura, H., Hasegawa, M., Murayama, S. and Mochizuki, H. (2022), TDP-43 Proteinopathy Presenting with Typical Symptoms of Parkinson's Disease. Mov Disord, 37: 1561-1563. link
- Jo, M., Lee, S., Jeon, YM. et al. The role of TDP-43 propagation in neurodegenerative diseases: integrating insights from clinical and experimental studies. Exp Mol Med 52, 1652–1662 (2020). https://doi.org/10.1038/s12276-020-00513-7 link
- Amador-Ortiz C, Lin WL, Ahmed Z, Personett D, Davies P, Duara R, Graff-Radford NR, Hutton ML, Dickson DW. TDP-43 immunoreactivity in hippocampal sclerosis and Alzheimer's disease. Ann Neurol. 2007 May;61(5):435-45. doi: 10.1002/ana.21154. PMID: 17469117; PMCID: PMC2677204. link
- Foulds P, McAuley E, Gibbons L, Davidson Y, Pickering-Brown SM, Neary D, Snowden JS, Allsop D, Mann DM. TDP-43 protein in plasma may index TDP-43 brain pathology in Alzheimer's disease and frontotemporal lobar degeneration. Acta Neuropathol. 2008 Aug;116(2):141-6. doi: 10.1007/s00401-008-0389-8. Epub 2008 May 28. PMID: 18506455; PMCID: PMC2464623. link
- Chen XQ, Barrero CA, Vasquez-Del Carpio R, Reddy EP, Fecchio C, Merali S, Deglincerti A, Fang C, Rogers J, Maccecchini ML. Posiphen Reduces the Levels of Huntingtin Protein through Translation Suppression. Pharmaceutics. 2021 Dec 7;13(12):2109. doi: 10.3390/pharmaceutics13122109. PMID: 34959389; PMCID: PMC8708689. link
Contacts
Maria Maccecchini
maccecchini@annovisbio.com