Innovia Medical Expands Single-Use Instrument Product Line into the US with Gynecology Product Cervical Rotating Biopsy Punch
Innovia Medical Expands Single-Use Instrument Product Line into the US with Gynecology Product Cervical Rotating Biopsy Punch
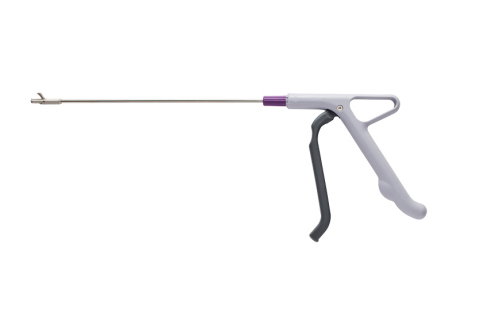
ST. PAUL, Minn.--(BUSINESS WIRE)--Innovia Medical (“Innovia”) is thrilled to announce the expansion of their single-use instrument product line into the U.S. The product leading the expansion is a gynecology product called the Cervical Rotating Biopsy Punch which supports urgently needed procedures like cancer diagnosis. The single-use instrument was first introduced by DTR Medical Ltd (“DTR Medical”) in Wales in 2012 is now offered by Summit Medical LLC. (“Summit Medical”), a Minnesota based manufacturer of medical devices.
Since the initial development of the Cervical Rotating Biopsy Punch, the product has been used by numerous National Health Service (NHS) clinics and hospitals in the U.K. that have provided annual product feedback allowing for continuous product innovations. As part of an ongoing project with a leading Welsh University, a pioneering technique, known as biorefining, was used to recreate low carbon technologies for both the Cervical Rotating Biopsy Punch and it’s packaging. The instrument handle is now made of 20% renewably sourced propanediol (PDO) made from corn starch, making it both lightweight and recyclable. The polybag packaging was biorefined in 2020 to save 1,200 pounds of plastic each year. The Cervical Rotating Biopsy Punch has 2.8 times less of an environmental impact than other single-use products on the market like the metal Tischler Cervical Biopsy Punch and Baby Tischler Cervical Biopsy Punch.
Single-use instruments like the Cervical Rotating Biopsy Punch eliminate cross contamination, inadequate samples and repeat procedures. With Cervical cancer screenings down 68% in the U.S. due to COVID-19 [1], the importance of an adequate biopsy is essential in reducing the time to an accurate diagnosis.
“We believe introducing the Cervical Rotating Biopsy Punch to the U.S. is a natural expansion of our Single-Use Instrument category. Immediate benefits will improve the quality of patient care from acute care facilities to small offices that lack reprocessing environments. The quality of biopsy is notable and user feedback over the past decade has been extremely positive.” Said Marcus Super, Vice President of Marketing at Innovia Medical.
Improving Patient Outcomes with Early Detection
Human Papillomaviruses (HPV) are the most widespread sexually transmitted infections (STI) worldwide with 12% of women having a detectable cervical HPV infection. HPV related cancer makes up 8.6% of all cancer cases worldwide [2]. When detected at an early stage, the 5-year survival rate for people with invasive cervical cancer in the U.S. is 92% [3]. Reducing the amount of time between diagnosis and treatment with adequate biopsies can improve patient outcomes.
The Cervical Rotating Biopsy Punch has been supported by Gynecologists with comments like “It’s amazing how light it is! I’m really impressed with the jaw, it’s so sharp” from Garima Srivastava, Gynecology Consultant at Harrogate District Hospital in the U.K.
The Cervical Rotating Biopsy Punch’s metal jaw is stronger than titanium and ensures a sharp, clean, 3.0 x 7.5mm cut that delivers consistent results even on postmenopausal women and patients with a hardened cervix leading to efficient Pathology laboratory results and quick diagnosis. The instrument’s low profile jaw ensures better access to the Transformation Zone when migrating into the endocervical canal. The 360-degree rotating head facilitates positioning and enables increased visibility.
Our 2019 survey conducted in the United Kingdom discovered that 88% of clinicians would recommend Innovia Medical’s Cervical Rotating Biopsy Punch to a colleague as a result of its high performance and effectiveness. The same survey found that 86% of clinicians described their first sample as either “good” or “very good” when asked about the quality of both the cut and the biopsy and the ease of removing the sample from the jaw. A high quality sample taken in just one attempt reduces cervical trauma and patient discomfort. Louise Johnson, Nurse Colposcopist at the University Hospital of Wales was quoted “Absolutely would recommend” when asked about the Biopsy Punch.
To learn more about the Cervical Biopsy Rotating Punch or Innovia Medical’s single-use instrument product line expansion into the U.S. visit www.innoviamedical.com.
Innovia Medical is a family of specialty surgical companies that are passionately committed to partnering with medical professionals to help elevate the delivery of patient care and improve clinical outcomes.
Innovia Medical companies develop and manufacture solutions designed with both the medical professional and their patients in mind, to deliver care that: saves time for both the medical professional and patient, delivers a consistent and predictable care experience, and improves patient confidence with reduced recovery times and less discomfort.
Innovia’s mission is to improve outcomes for the global healthcare community and the patients they serve through innovative solutions that elevate the delivery of care.
Summit Medical, headquartered in St. Paul, Minnesota, is a medical device manufacturer that has been serving the global healthcare community for over 35 years. The company is dedicated to staying on top of industry demands and developing solutions to meet them through all the stages of design, engineering, and manufacturing.
Summit Medical works closely with surgeons, nurses, and allied healthcare professionals to develop unique, patented products that improve surgical outcomes and make the delivery of healthcare more efficient. Most Summit Medical products are manufactured and packaged in-house and sold by a global network of independent specialty distributors that are supported with a complete marketing program.
DTR Medical, based in Swansea, United Kingdom, specializes in the design and manufacture of sterile single-use surgical instruments that deliver both patient and clinical value. Exporting to over 30 countries they have built their award-winning reputation on engineering high-quality, innovative products whilst maintaining exceptional customer service.
Since their founding in 2005, DTR Medical has been dedicated to providing products designed to the highest specification that are in line with their core value of Innovation, Quality and Service.
References
[1] Respaut, R., & Nelson, D. J. (2020, April 28). Exclusive: U.S. Medical Testing, cancer screenings plunge during coronavirus outbreak - data firm analysis. Reuters. Retrieved December 3, 2021, from https://www.reuters.com/article/us-health-coronavirus-usa-screenings-exc/exclusive-u-s-medical-testing-cancer-screenings-plunge-during-coronavirus-outbreak-data-firm-analysis-idUSKCN22A0DY.
[2] de Martel, C., Plummer, M., Vignat, J., & Franceschi, S. (2017). Worldwide burden of cancer attributable to HPV by site, country and HPV type. International journal of cancer, 141(4), 664–670. Retrieved December 3, 2021, from https://doi.org/10.1002/ijc.30716.
[3] American Society of Clinical Oncology. (2021, February 8). Cervical cancer - statistics. Cancer.Net. Retrieved December 3, 2021, from https://www.cancer.net/cancer-types/cervical-cancer/statistics.
Contacts
Lauren Cook
651.789.8166
media@innoviamedical.com