SpotSee Launches WarmMark Indicator with Additional Time and Temperature Thresholds to Match COVID-19 Vaccines
SpotSee Launches WarmMark Indicator with Additional Time and Temperature Thresholds to Match COVID-19 Vaccines
Indicators will be used to ensure temperature and time tolerances are maintained during transport and prior to administration of COVID-19 vaccine
DALLAS--(BUSINESS WIRE)--SpotSee®, a global leader in supply chain temperature indicators used to monitor test specimens in the SARS and H1N1 virus outbreaks, has introduced new temperature thresholds for its WarmMark indicators to address temperature requirements associated with the vaccine for the coronavirus disease 2019 (COVID-19).
“In the rush to get vaccines developed and distributed to vaccination sites, there hasn’t been much attention paid to the last 10 feet after the vaccines have thawed at the administration site, where the opportunity for vaccine efficacy erosion due to temperature exposure is greatest,” said Tony Fonk, CEO, SpotSee. “Our indicators help administrators in hospitals, nursing homes, retail pharmacies and other vaccination sites know if a vaccine has exceeded its temperature threshold over time. Our products are well known for similar use in annual flu vaccinations, and we have customized our indicator time and temperature requirements to match the COVID-19 vaccine efficacy thresholds.”
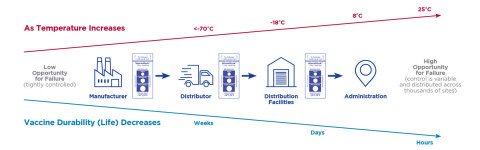
According to the Centers for Disease Control and Prevention’s (CDC) guiding principles, during a pandemic, efficient, expeditious and equitable distribution and administration of approved vaccine is critical. Additionally, it states that vaccine safety standards will not be compromised in efforts to accelerate COVID-19 vaccine development or distribution. The COVID-19 vaccine, regardless of manufacturer, must maintain a specific temperature threshold or else the vaccine is rendered ineffective. Each temperature threshold is different but can range from -70°C to 8°C depending on if the vaccine is being distributed, stored or thawed for use. All COVID-19 vaccine candidates have some limited time period where they can be stored in a refrigerated environment (2°C to 8°C) before administering to patients.
SpotSee’s WarmMark indicator is used at various times throughout the supply chain of vaccines. Some major vaccine manufacturers apply the indicator at the point of production to vaccine vial boxes or place the indicator inside. In other cases, large pharmaceutical distributors add the indicators as they break down bulk shipments into smaller, insulated shippers. One of the largest applications of the WarmMark indicators is at vaccine administration sites, after a shipment of vaccines has been received. The vaccines are exposed to different temperatures as refrigerators are opened and closed and vaccines travel back and forth in the “last 10 feet” before injection. This is where most temperature-related efficacy and safety issues can occur. Using the time and temperature feature, administrators will know if doses have experienced unacceptable deviations from the predetermined temperature thresholds.
The WarmMark temperature indicator is available in different temperature sensitivities ranging from -18°C/0°F to 37°C/99°F, with specific requirements for acceptable out-of-refrigeration periods. The new variants include both 8°C and 25°C versions with shorter run-out times. The new time and temperature ranges are as follows:
- 8°C / 6 hours
- 8°C / 14 days
- 25°C / 6 hours
- 2°C / 48 hours
The indicator can be applied to COVID-19 packaging before it is shipped or stored to maintain the vaccine’s efficacy throughout the supply chain. This low-cost solution provides a clear, easy-to-read indication so healthcare workers can be completely confident when vaccinating patients.
SpotSee’s temperature indicators are used throughout the world to ensure accurate results for shipping and administration of various vaccines, biologics and specimens.
To learn more about WarmMark and its capabilities, or to purchase these indicators for COVID-19 vaccine monitoring, visit: https://bit.ly/3oySXhA.
About SpotSee
SpotSee is an end-to-end solution provider that enables customers to spot damage in their operations and see it in real time. SpotSee’s products are used to monitor the condition of everything from vaccines to spaceships. SpotSee has a full range of visual and connected devices that deliver data from devices anywhere in the world to customers’ fingertips. SpotSee devices monitor shock, vibration, temperature and other environmental conditions through its market-leading brands such as ShockWatch®, ShockLog®, SpotBot™, OpsWatch and WarmMark®. SpotSee’s temperature indicators have been instrumental in monitoring vaccines for more than 20 years, including those for SARS, H1N1 and seasonal flu, to ensure the cold chain is maintained. To address the needs of the global community during the coronavirus pandemic, SpotSee has donated indicators for test specimen monitoring and updated products to ensure COVID-19 vaccine temperature monitoring. The company has a global network of over 300 sales and technical service partners in 62 countries. SpotSee is headquartered in Dallas, Texas, with operations in Brazil, Netherlands, United Kingdom, China, Mexico and Graham, Texas. For more information, visit www.spotsee.io.
Contacts
Elle Glatz
Mod Op for SpotSee
Elle.glatz@modop.com