KARL STORZ Receives FDA Clearance for FIVE S 6.5 Bronchoscope
KARL STORZ Receives FDA Clearance for FIVE S 6.5 Bronchoscope
High-suction device is designed to maximize control, clarity, and confidence in critical settings.
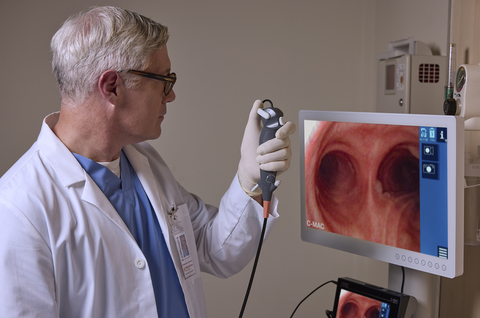
EL SEGUNDO, Calif.--(BUSINESS WIRE)--KARL STORZ, a global leader in endoscopic and minimally invasive surgical technology, has received FDA clearance for the FIVE S 6.5 sterile single-use bronchoscope. The device offers a valuable new option for critical care physicians, intensivists, RTs, and ICU-based proceduralists seeking airway control solutions for high-risk patients—especially those managing heavy secretions or in urgent situations requiring fast airway clearance.
KARL STORZ announces FDA clearance for the FIVE S 6.5 sterile single-use bronchoscope.
Share
“The FIVE S 6.5 single-use bronchoscope delivers precise articulation, powerful secretion clearance, and sharp visualization,” said Jennifer Jackson, Executive Director, Sales & Marketing, Airway and Emergency Medicine at KARL STORZ North America. “Whether performing bronchoscopy, lavage, or airway inspection, the FIVE S 6.5 provides the reliability you need when every second counts.”
The FIVE S 6.5 bronchoscope delivers powerful suction, thanks to a large 3.0 mm working channel. During bench testing, the device cleared 15 mL of simulated mucus (prepared using egg whites) in less than 5 seconds.
Designed to perform like a reusable scope, the FIVE S 6.5 incorporates numerous user-centered features that are vital in critical care and sure to impress healthcare providers. For example, the bronchoscope features a reinforced shaft and specialized tip designed to support controlled navigation and scope positioning in challenging airways. The contoured grip and balanced weight are designed to support maneuverability and operator comfort during extended procedures. Thanks to seamless integration with the industry-leading C-MAC™ monitor, users can expect consistent illumination and crystal-clear image quality during every procedure. Support for accessories up to 2.4 mm outer diameter permits use with both single-use and reusable instruments for procedural flexibility.
Customers who currently use the C-MAC™ video laryngoscopy system will find it easy to add the FIVE S 6.5 bronchoscope, which uses just a single connection cable, thereby minimizing both training and setup time. The shared visualization platform supports both bronchoscopy and intubation workflows to maximize value. Cross-compatibility with single-use FIVE S bronchoscopes and reusable or single-use C-MAC™ video laryngoscopes further advances procedural flexibility.
“At KARL STORZ, we understand that managing costs goes far beyond purchase price and encompasses workflow efficiency, reliability, performance, reprocessing, and infection control,” Jackson said. “With the FIVE S 6.5—as with the entire FIVE product family—we help our partners address all of these concerns though a single cost-effective, low-maintenance, contamination-reducing solution.”
To learn more about the FIVE S 6.5 and related airway management solutions, visit karlstorz.com.
About KARL STORZ
KARL STORZ is an innovative leader in endoscopic technology and surgical imaging across virtually all surgical specialties. Its diverse portfolio of airway management solutions sets the standard for performance, reliability, simplicity, and cross-compatibility across all sites of care. Based in Germany, KARL STORZ is a global family-owned company that designs, engineers, manufactures, and markets all its products with an emphasis on visionary design, precision craftsmanship, and clinical effectiveness. For more information, visit www.karlstorz.com.
Contacts
Media contact:
Susan Mancia
Marketing Communications
KARL STORZ North America
657-385-5285