Stratasys Expands Availability of RadioMatrix™ Radiopaque Material to the United States
Stratasys Expands Availability of RadioMatrix™ Radiopaque Material to the United States
First-ever radiopaque 3D printing material now fully available to U.S. healthcare providers
MINNETONKA, Minn. & REHOVOT, Israel--(BUSINESS WIRE)--Stratasys Ltd. (NASDAQ: SSYS) today announced full commercial availability of its RadioMatrix™ radiopaque 3D printing material in the United States. This milestone follows initial limited deployments and marks the first time healthcare providers, device manufacturers, and research institutions across the U.S. can broadly access and utilize the material for advanced medical imaging and training applications.
RadioMatrix is the first and only 3D printing material that enables precise control of radiopacity - allowing users to produce patient-specific models with repeatable, consistent, and fully tunable visibility on X-ray based imaging. Stratasys developed RadioMatrix to support a new era of medical imaging. Its unmatched fidelity for computed tomography (CT) phantoms was later underscored by research conducted with Siemens Healthineers, which validated RadioMatrix’s capabilities and accuracy for accelerating innovation in device testing, calibration, and education.
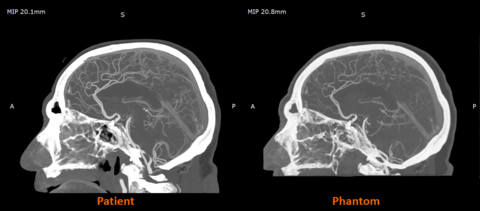
UK-based work with partners such as CPI and Beaumont Hospital is already demonstrating the impact of radiopaque 3D-printed models in practice, with radio-realistic cerebral angiography phantoms being used to improve the fidelity of imaging-based training and create more controlled, repeatable environments for research.
Early research from the Stratasys–Siemens Healthineers collaboration shows that 3D printed RadioMatrix™ phantoms can closely replicate real human tissue in CT imaging, with deviations reported as low as single Hounsfield units (HU) in critical areas such as grey matter and veins. By combining Stratasys’ Digital Anatomy™ 3D printing technology and radiopaque materials with advanced imaging algorithms, the partners are demonstrating anatomically realistic, radio-accurate phantoms that preserve fine anatomical details and pathological variations while offering a more consistent, ethical alternative to cadavers. These models are expected to improve how radiologists validate and optimize CT protocols and accelerate the development of new imaging algorithms for more precise diagnosis and treatment planning.
“Providing full availability of RadioMatrix in the U.S. is a major step in providing cutting-edge imaging education and training,” said Erez Ben Zvi, Vice President, Healthcare, Stratasys. “By giving radiologists and device manufacturers the ability to print ultra-realistic, customized radiographically accurate models, we’re helping replace traditional phantom solutions and reliance on cadavers with customizable, repeatable, and scalable alternatives.”
For more information or to request a benchmark model, contact a Stratasys representative or visit http://www.stratasys.com/healthcare.
About Stratasys
Stratasys is leading the global shift to additive manufacturing with innovative 3D printing solutions for industries such as aerospace, automotive, consumer products, and healthcare. Through smart and connected 3D printers, polymer materials, a software ecosystem, and parts on demand, Stratasys solutions deliver competitive advantages at every stage in the product value chain. The world’s leading organizations turn to Stratasys to transform product design, bring agility to manufacturing and supply chains, and improve patient care.
To learn more about Stratasys, visit www.stratasys.com, the Stratasys blog, X/Twitter, LinkedIn, or Facebook. Stratasys reserves the right to utilize any of the foregoing social media platforms, including Stratasys’ websites, to share material, non-public information pursuant to the SEC’s Regulation FD. To the extent necessary and mandated by applicable law, Stratasys will also include such information in its public disclosure filings.
Contacts
Media and Investor contacts:
Stratasys Corporate, North America & EMEA
Chris Reese
chris.reese@stratasys.com
+1 651 357 0877
Stratasys Corporate, Israel & EMEA
Erik Snider
Erik.Snider@stratasys.com
+972 74 745 6053
Investor Relations
Yonah Lloyd
Yonah.Lloyd@stratasys.com
+972 74 745 4919