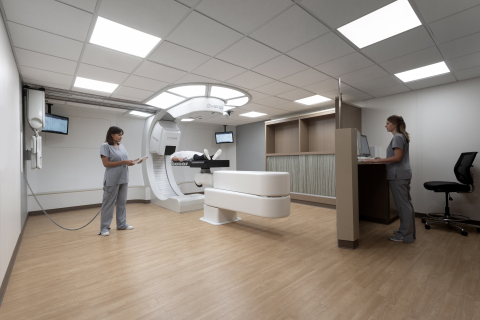
Mevion Selected to Equip Compact Proton Therapy Center in Kansas City
Mevion Selected to Equip Compact Proton Therapy Center in Kansas City
The physician-led center will have the first Mevion compact proton therapy system in Kansas
LITTLETON, Mass,--(BUSINESS WIRE)--Mevion Medical Systems is pleased to announce it has signed a contract with Kansas City Urology Care (KCUC) for the purchase of a MEVION S250i Proton Therapy System with HYPERSCAN Pencil Beam Scanning technology.
Proton therapy is a precise form of radiation therapy that can reduce the amount of unnecessary radiation exposure to healthy tissue, which has the potential to reduce side effects, escalate dose, and lessen the risk of secondary malignancies. HYPERSCAN technology utilizes a unique combination of optimum spot sizes, hyper-fast layer switching and a novel multi-layer proton MLC. This powerful trio provides faster and more precise dose delivery and minimizes sensitivity to motion, allowing clinicians to deliver a full field of therapeutic radiation in a single breath-hold.
The size and cost of larger proton technologies has been a major impediment to the adoption of proton therapy. By reducing the size and complexity of a proton therapy system, Mevion has overcome the practical challenges of other technologies, and created a much more compact, affordable solution. Today, new proton therapy centers in the US are almost exclusively compact single-room systems and Mevion is the preferred compact provider in the US.
“Mevion has taken the lead and has democratized proton therapy to the point where centers of all sizes can participate in the incredible benefits proton therapy can provide to their patients,” said Tina Yu, CEO of Mevion. “We are very excited to partner with KCUC to bring this technology to the patients of Kansas City.”
Kansas City Urology Care is the premier center for urology care in Kansas City that assures cutting-edge treatment solutions for all forms of urological conditions. For more, information about Kansas City Urology Care, please visit, www.KCUC.com.
2021 ASTRO Annual Meeting
Mevion will attend the American Society of Radiation Oncology (ASTRO) 2021 Annual Meeting from October 24th-26th. After 2020’s virtual-only event, ASTRO 2021 returns live at McCormick Place in West Chicago, where Mevion will highlight its recent updates, including a FLASH Therapy* research kit, that is expected to be made available to users in 2022, to help empower research centers of all sizes to study the potential benefits of FLASH. Mevion also invite attendees to experience the speed and precision of HYPERSCAN with RayStation 11A and learn more about our 24/7 clinical availability and the fastest installation time in the industry at booth #601.
*FLASH Therapy is currently under preclinical research and is not yet available for commercial sale or clinical use.
To receive updates during ASTRO:
- Follow us on Twitter: @MevionMedical
- Connect with us on LinkedIn: Mevion Medical Systems
- Like Us on Facebook: MevionMedical
About Mevion Medical Systems
Since 2004, Mevion Medical Systems has been the leading provider of compact proton therapy systems for cancer care. Dedicated to advancing the design and accessibility of proton therapy worldwide, Mevion was the first company to innovate this new single-room platform and continues to further the science and application of proton therapy. Mevion’s flagship product, the MEVION S250i Proton Therapy System with HYPERSCAN pencil beam scanning, is the world’s smallest proton therapy system that eliminates the obstacles of size, complexity, and cost that exists with other proton therapy systems. Mevion is headquartered in Littleton, Massachusetts, with a presence in Europe and Asia. For more information, please visit www.mevion.com.
Contacts
Melanie Benton
Director, Marketing
978-540-1551
mbenton@mevion.com