Iveric Bio Announces Presentation of Post-Hoc Analysis from GATHER1 Clinical Trial of Zimura® in Patients with Geographic Atrophy
Iveric Bio Announces Presentation of Post-Hoc Analysis from GATHER1 Clinical Trial of Zimura® in Patients with Geographic Atrophy
- Analysis to be Presented at Retinal World Congress in Fort Lauderdale, FL on May 13, 2022 -
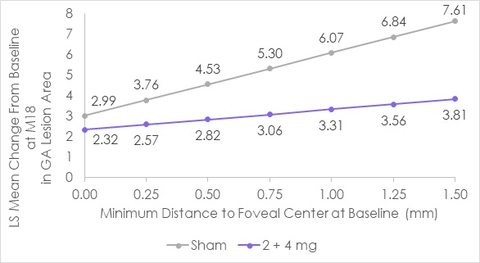
PARSIPPANY, N.J.--(BUSINESS WIRE)--IVERIC Bio, Inc. (Nasdaq: ISEE) announced today that a post-hoc analysis evaluating the reduction in geographic atrophy (GA) lesion growth observed in patients receiving Zimura® (avacincaptad pegol) as compared to patients receiving sham in the completed GATHER1 clinical trial will be presented at the upcoming Retinal World Congress in Fort Lauderdale, FL on May 13, 2022. The new results analyze the reduction in GA lesion growth, in a subset of GATHER1 patients, based on the distance of a patient’s GA lesion from the foveal center at baseline.
In-line with the overall results of GATHER1, a reduction of GA lesion growth in patients receiving Zimura as compared to patients receiving sham was consistently observed across all baseline distances from the foveal center. No additional safety analysis was performed as part of the post-hoc analysis.
“Analyses from other GA studies have shown that lesions farther away from the foveal center grow faster than lesions closer to the foveal center,” stated Dhaval Desai, PharmD, Chief Development Officer of Iveric Bio. “In addition to supporting these findings, importantly a reduction in lesion growth for GA patients receiving an investigational product was observed in distances ranging from 250 µm to 1500 µm from the foveal center. However, the greatest reduction in GA growth was observed among the group of patients with lesions farthest from the foveal center at baseline.”
Glenn J. Jaffe, MD, Director, Duke Reading Center, Chief, Retina Division, Duke Eye Center, Robert Machemer Professor of Ophthalmology added, “These results suggest that early administration with Zimura to patients with GA lesions, when the lesion is still farther away from the foveal center and is growing the fastest, may be most beneficial.”
The accompanying graphs summarize the results of this retrospective review.
The analysis will be presented at Retinal World Congress in Fort Lauderdale, FL. Details of the presentations are as follows:
Presentation Title: Evaluation of GA Lesion Growth by Minimum Distance to the Fovea Center: Post-Hoc Analysis of the GATHER1 Trial
- Presenter: Dr. Glenn Jaffe
- Date / Time: Friday, May 13, 2022 at 5:30 PM (ET)
Iveric Bio will make the full set of slides of the presentation available on the Company’s website at https://investors.ivericbio.com/events-and-presentation at the start of the presentation.
About GATHER1 and GATHER2
The Company previously announced that in GATHER1, Zimura (avacincaptad pegol) met its pre-specified primary efficacy endpoint with statistical significance. The most frequently reported ocular adverse events in this trial were related to the injection procedure. The Company expects topline data for GATHER2, a second Phase 3 clinical trial for Zimura for GA, to be available in the third quarter of 2022, approximately one year after the enrollment of the last patient in the trial plus the time needed for database lock and analysis. If 12-month results from GATHER2 are positive, the Company plans to submit applications with the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for marketing approval of Zimura for GA. There are no FDA or EMA approved treatments available for patients with GA.
About Zimura
Zimura (avacincaptad pegol) is an investigational drug product and has not been approved for use anywhere globally. Zimura is designed to target and inhibit the cleavage of complement protein C5 and the formation of its downstream fragments, C5a and C5b. By inhibiting the formation of these fragments, Zimura is believed to decrease or slow the chronic inflammation and cell death associated with the retinal aging process by decreasing the formation of membrane attack complex (MAC) and inflammasome activity, thereby potentially avoiding or slowing the degeneration of retinal pigment epithelial cells. This potential mechanism is the rationale for Zimura as a potential therapy for geographic atrophy.
About Iveric Bio
Iveric Bio is a science-driven biopharmaceutical company focused on the discovery and development of novel treatments for retinal diseases with significant unmet medical needs. The Company is committed to having a positive impact on patients’ lives by delivering high-quality, safe and effective treatments designed to address debilitating retinal diseases including earlier stages of age-related macular degeneration. For more information on the Company, please visit www.ivericbio.com.
Forward-looking Statements
Any statements in this press release about Iveric Bio’s future expectations, plans and prospects constitute forward-looking statements for purposes of the safe harbor provisions under the Private Securities Litigation Reform Act of 1995. Forward-looking statements include any statements about the Company’s strategy, future operations and future expectations and plans and prospects for the Company, and any other statements containing the words “anticipate,” “believe,” “estimate,” “expect,” “intend”, “goal,” “may”, “might,” “plan,” “predict,” “project,” “seek,” “target,” “potential,” “will,” “would,” “could,” “should,” “continue,” and similar expressions. In this press release, the Company’s forward looking statements include statements about its expectations regarding its development and regulatory strategy for Zimura, including the timing of receipt of topline data from the GATHER2 clinical trial and its plans to file for marketing approval for geographic atrophy if the results of GATHER2 are positive, the potential utility of Zimura and the clinical meaningfulness of clinical trial results and data, including from post-hoc analyses of the GATHER1 clinical trial. Such forward-looking statements involve substantial risks and uncertainties that could cause the Company’s development programs, future results, performance, or achievements to differ significantly from those expressed or implied by the forward-looking statements. Such risks and uncertainties include, among others, those related to the progress and success of research and development programs and clinical trials, developments from the scientific and medical community and other factors discussed in the “Risk Factors” section contained in the quarterly and annual reports that the Company files with the Securities and Exchange Commission. Any forward-looking statements represent the Company’s views only as of the date of this press release. The Company anticipates that subsequent events and developments may cause its views to change. While the Company may elect to update these forward-looking statements at some point in the future, the Company specifically disclaims any obligation to do so except as required by law.
ISEE-G
Contacts
Investor Contact:
Kathy Galante
Senior Vice President, Investor Relations
kathy.galante@ivericbio.com
or
Media Contact:
Jeannie Neufeld
Senior Director, Public Relations & Communications
jeannie.neufeld@ivericbio.com