ROCKVILLE, Md.--(BUSINESS WIRE)--BioMarker Strategies LLC today announced that the Korean Patent Office has granted a new patent covering its PathMAP® Functional Signaling Profile technology.
“PathMAP Functional Signaling Profiles are highly predictive of individual solid tumor responses to targeted therapies and combinations because they are based on the dynamic, predictive signaling information available only from live cells,” said Jerry Parrott, President and CEO, BioMarker Strategies.
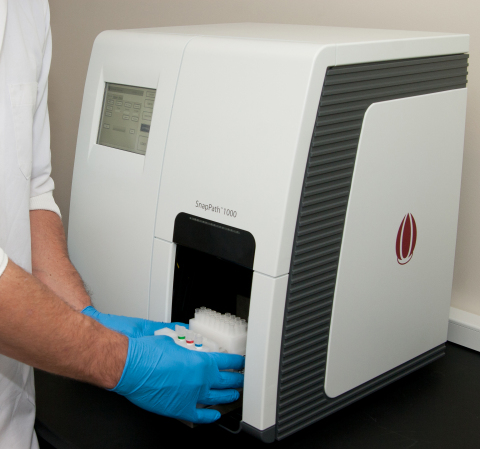
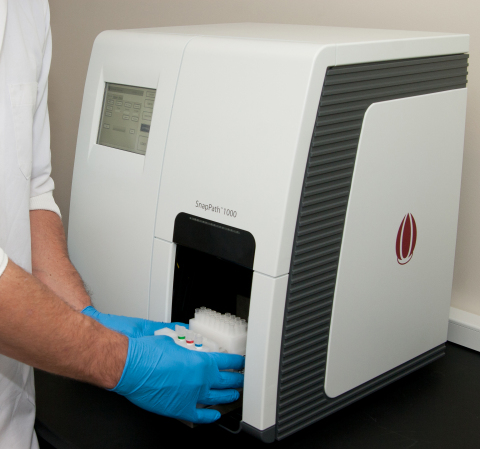
PathMAP Functional Signaling Profiles are made possible by the Company’s proprietary SnapPath® Cancer Diagnostics System, which was developed specifically to enable predictive tests to guide targeted drug development and treatment selection for patients with solid tumor cancers. SnapPath is the only diagnostics system that can generate purified populations of live solid tumor cells from live, unfixed samples in an automated and standardized manner. The system preserves the molecular integrity of these living cells for ex vivo exposure to targeted therapies on the SnapPath System. The Korean Patent Office previously granted a patent covering the Company’s SnapPath® Cancer Diagnostics System technology in 2018.
“Patents on both of our core technologies have now been granted in the United States, Europe, Australia, Canada, Japan and Korea,” said Mr. Parrott. “These and other granted patents give us broad protection to advance our mission to enable the use of live cells to personalize drug development and treatment selection for patients with solid tumor cancers.”
Patents Granted BioMarker Strategies for Its SnapPath® Cancer Diagnostics System: “Improved Methods and Devices for Cellular Analysis”
|
Country/Region |
Patent # |
Issue Date |
Expiration |
||||||||||
| United States | US9121801 | September 1, 2015 | December 28, 2030 | ||||||||||
| Europe | EP2210080 | January 28, 2015 | October 24, 2028 | ||||||||||
| Australia | AU2008317329 | January 15, 2015 | October 24, 2028 | ||||||||||
| Australia | AU2014277688 | January 19, 2017 | October 24, 2028 | ||||||||||
|
Canada |
CA2703631 |
August 22, 2017 |
October 24, 2028 |
||||||||||
| Hong Kong | HK1145878 |
August 14, 2015 |
October 24, 2028 |
||||||||||
| Japan | JP6177841 | July 21, 2017 | October 24, 2028 | ||||||||||
| Korea | KR10-1658245 | September 9, 2016 | October 24, 2028 | ||||||||||
Patents Granted for the Company’s PathMAP® Functional Signaling Profile Technology: “Compositions for Prediction of Drug Sensitivity, Resistance and Disease Progression”*
|
Country/Region |
Patent # |
Issue Date |
Expiration |
||||||||||
| United States | US9766249 | September 19, 2017 | August 31, 2025 | ||||||||||
| Europe | EP2561368 | August 2, 2017 | April 18, 2031 | ||||||||||
| Australia | AU2011242990 | May 28, 2015 | April 18, 2031 | ||||||||||
| Canada | CA2795362 | March 20, 2018 | April 18, 2031 | ||||||||||
|
China |
ZL201180030332.9 |
April 13, 2018 | April 18, 2031 | ||||||||||
| Japan | JP6158078 | June 16, 2017 | April 18, 2031 | ||||||||||
| Korea | KR10-1977875 | May 7, 2019 | April 18, 2031 | ||||||||||
| Singapore | SG184507 | May 6, 2015 | April 18, 2031 | ||||||||||
The SnapPath and PathMAP technologies are ideally suited to assess response to targeted drugs and combinations, including combinations with Immunotherapies, in development for the treatment of solid tumor cancers.
About BioMarker Strategies
BioMarker Strategies is a privately-held molecular diagnostics company founded in 2006 at The Johns Hopkins Science & Technology Park in Baltimore, and now based in Rockville, Maryland. The Company’s mission is to help make personalized medicine more of a reality for patients with solid tumor cancers, by providing powerful new molecular tools and research services to guide targeted drug development and treatment selection for these patients. For more information about BioMarker Strategies, please see www.biomarkerstrategies.com.
Forward-Looking Statements
The information in this press release includes our projections and other forward-looking statements regarding future events. In some cases, forward-looking statements may be identified by terminology such as “may,” “will,” “should,” “expects,” “intends,” “plans,” “anticipates,” “believes,” “projects,” “estimates,” “predicts,” “potential,” “continue”, etc. These statements are not guarantees of future performance or achievement and involve certain risks and uncertainties, which are difficult to predict. Therefore, actual future results and trends may differ materially from what is projected here.