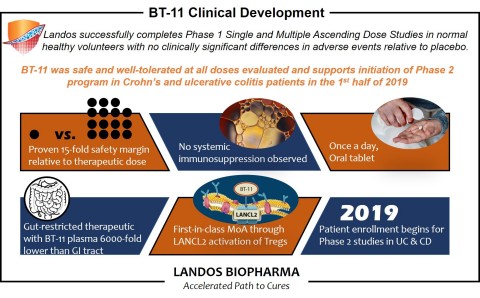
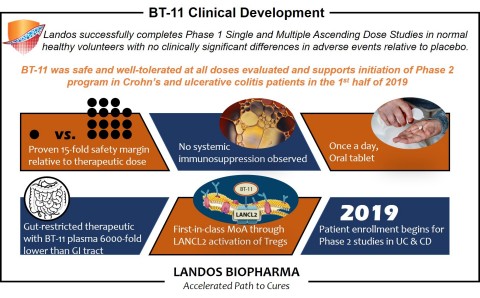
BLACKSBURG, Va.--(BUSINESS WIRE)--Landos Biopharma, Inc., an emerging clinical-stage, biopharmaceutical company focused on developing improved treatments for autoimmune diseases, today announces final results from the Phase 1 study of BT-11, a first-in-class, orally active, gut-restricted therapeutic for the treatment of ulcerative colitis (UC) and Crohn’s disease (CD).
The randomized, double-blind, placebo-controlled trial conducted in 70 healthy subjects to assess the safety of BT-11 found that BT-11 was well-tolerated and showed no dose-limiting toxicities. Among the subjects, the overall adverse events (AEs) profile observed was consistent when compared to AEs in the placebo cohort and no differences were observed between groups. The study tested levels of BT-11 ranging from 7 mg/kg up to 100 mg/kg and did not reach a maximum tolerated dose.
“The results of our Phase 1 study provide the first-in-human clinical data in support of BT-11 as a novel oral treatment for IBD. We are pleased to see that BT-11 was well-tolerated and safe in healthy volunteers,” said Dr. Josep Bassaganya-Riera, Chairman and CEO of Landos. “Importantly, we are very encouraged by the reduction of fecal calprotectin in BT-11-treated subjects, which could represent an early anti-inflammatory signal, and supports the potential of BT-11 as an important advancement in the treatment of inflammatory bowel disease. These study results, along with extensive preclinical efficacy models of IBD, support the evaluation of BT-11 in Phase 2 proof-of-concept efficacy studies in UC and CD patients, which we plan to initiate this year.”
“These results are promising for IBD patients in need of a more convenient treatment for UC and CD,” said Jean-Frederic Colombel, MD, a Landos Clinical Advisory Board member, world-renowned gastroenterologist and Director of the IBD Center at the Icahn School of Medicine at Mount Sinai. “We continue to see an unmet clinical need for chronic oral therapies to treat UC and CD that can improve safety, tolerability and convenience.”
About the Phase 1 Trial
This randomized, double-blind,
placebo-controlled Phase 1 trial evaluated the safety and tolerability
of BT-11 in five cohorts of single ascending and three cohorts of
multiple ascending dose studies in 70 healthy subjects. The study tested
levels of BT-11 ranging from 7 mg/kg up to 100 mg/kg. Within the subject
group of the study, BT-11 was well-tolerated and showed no safety
concerns or dose-limiting toxicities.
In addition, biomarker and immune screening results indicated that subjects in the single and multiple ascending dose studies had lower levels of fecal calprotectin, an inflammatory biomarker, compared to the placebo cohort. In addition, BT-11 did not induce systemic immunosuppression as measured by white blood cell counts relative to placebo in either study.
There were no dose dependent relationships in gastrointestinal-related AEs observed in both the single and multiple ascending dose studies. There was no significant difference in AEs between the dosed subjects and the placebo cohort.
About BT-11
Landos’ lead clinical asset, BT-11, is a novel,
oral, gut-restricted, small molecule targeting the Lanthionine
Synthetase C-Like 2 (LANCL2) pathway in the gastrointestinal tract for
the treatment of Crohn’s disease (CD) and ulcerative colitis (UC). BT-11
has shown outstanding therapeutic efficacy in preclinical models of
inflammatory bowel disease (IBD), a benign safety profile without the
concerns of systemic exposure, and has two open Investigational New Drug
(INDs) for evaluation in UC and CD. BT-11 intercepts IBD by decreasing
the production of inflammatory mediators and increasing
anti-inflammatory molecules within the gastrointestinal tract.
About IBD
IBD represents a group of chronic and
disabling disorders that greatly impact a patient’s quality of life. The
two primary clinical manifestations of IBD - Crohn’s disease (CD) and
ulcerative colitis (UC) - afflict 2 million North Americans and 5
million people worldwide, with nearly 25% growth in prevalence over the
last five years. There is an unmet clinical need for safer, more
effective medications for these diseases as currently marketed
therapeutics have a number of drawbacks: they only benefit a small
number of the overall population, lose response effectiveness, or cause
high rates of serious side effects, including cancer, infection, and
death.
About Landos Biopharma
Landos Biopharma, Inc. is a
clinical-stage biopharmaceutical company focused on the discovery and
development of first-in-class oral therapeutics for patients with
autoimmune diseases. Landos’ lead clinical asset, BT-11, is a
first-in-class, oral therapeutic that acts locally in the
gastrointestinal tract for treatment of inflammatory bowel disease
(IBD). The company has completed Phase 1 clinical testing and plans to
initiate Phase 2 clinical testing of BT-11 for inflammatory bowel
disease in 2019. Landos also has a robust pipeline of new compounds for
other autoimmune diseases, several of which will advance to IND in 2019.
Landos is headquartered in Blacksburg, VA. For more information, please
visit www.landosbiopharma.com or
contact info@landosbiopharma.com or
follow us @Landosbio.