CAMBRIDGE, Mass.--(BUSINESS WIRE)--Ra Pharmaceuticals, Inc. (NASDAQ: RARX) today announced positive interim results from the Company’s ongoing, global Phase 2 clinical program evaluating RA101495 SC for the treatment of paroxysmal nocturnal hemoglobinuria (PNH). Ra Pharma is a clinical stage biopharmaceutical company focusing on the development of next-generation therapeutics for the treatment of complement-mediated diseases and is developing RA101495 as a novel, subcutaneously-administered (SC) inhibitor of complement component 5 (C5).
The global, dose-finding Phase 2 clinical program is designed to evaluate the safety, tolerability, preliminary efficacy, pharmacokinetics (PK), and pharmacodynamics (PD) of RA101495 SC in patients with PNH. As of the data cut-off date of November 30, 2017, a total of 29 patients have been dosed with RA101495 SC in three cohorts:
- Eculizumab naïve: 10 patients enrolled, dosing completed
- Eculizumab switch: 16 patients enrolled, dosing ongoing
- Eculizumab inadequate responders: 3 patients enrolled, dosing ongoing
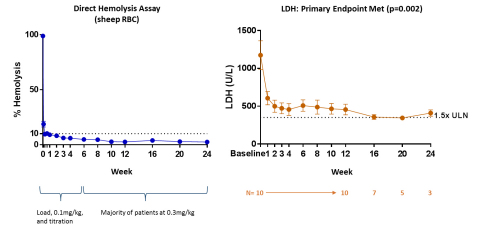
Eculizumab Naïve Cohort: RA101495 SC met the primary endpoint in eculizumab naïve patients (n=10). In these patients, a rapid, robust, and sustained reduction in lactate dehydrogenase (LDH) levels from baseline to the mean of Weeks 6-12 (p=0.002) and near-complete suppression of complement activity were observed (See figure). Fifty percent of eculizumab naïve patients who were transfusion-dependent prior to enrollment have been transfusion-free since commencing RA101495 SC. Based on preliminary data, meaningful improvements in standard measures of quality of life, as shown by the Functional Assessment of Chronic Illness Therapy (FACIT) fatigue score, have been observed, as well as a high level of patient satisfaction with subcutaneous self-administration based on patient surveys completed to date. Furthermore, 8 of 10 subjects enrolled in the study have elected to continue treatment with RA101495 SC in a long-term extension study.
Eculizumab Switch Cohort: Interim results from the ongoing switch cohort demonstrate near complete, sustained, and uninterrupted inhibition of complement activity during and after eculizumab washout. The LDH response observed in switch patients is bimodal based on prior transfusion requirements on eculizumab. In transfusion-independent patients from this cohort (n=5), a population segment representing approximately 80% of patients on long-term eculizumab therapy, switching to RA101495 SC has been successful as indicated by stable LDH levels and no episodes of breakthrough hemolysis. Among difficult to treat eculizumab patients who were transfusion-dependent at baseline (n=11) from this cohort, breakthrough hemolysis occurred after switching in 7 patients, who all reverted to eculizumab without complications.
Inadequate Responder Cohort: In the U.S.-based cohort of inadequate responders to eculizumab, who have a history of elevated LDH, 3 patients have been enrolled. LDH stabilization and relief of side effects associated with eculizumab intolerance have been observed in the first patient enrolled in this cohort.
Across all cohorts, no meaningful safety or tolerability concerns have been identified after more than 300 patient weeks of cumulative exposure. The majority of adverse events were deemed unrelated to the study drug and the most frequent study drug-related adverse event to date was headache. No meningococcal infections or thromboembolic events have been observed. Out of more than 2,000 doses administered, only 7 mild (grade 1) injection site reactions have occurred among 3 patients. Full compliance with once daily subcutaneous self-administration of RA101495 SC has been observed thus far.
“These results support the potential for RA101495 to be a safe, effective, and convenient subcutaneous, self-administered therapeutic option that patients may readily adopt,” said Principal Investigator Anita Hill, M.D., PhD, MRCP, FRCPath, Consultant Haematologist for Leeds Teaching Hospitals NHS Trust, UK, and Lead Clinician for the National PNH Service in England. “This is evident from the meaningful and sustained LDH reduction seen in treatment naïve patients, and by maintenance of LDH control in transfusion-independent switch patients, who make up the majority of the eculizumab-treated population.”
“RA101495 has now demonstrated the potential to serve as first-line therapy for newly diagnosed patients with PNH, as well as an attractive alternative for transfusion-free patients switching from eculizumab,” said Doug Treco, PhD, President and Chief Executive Officer of Ra Pharma. “We believe the vast majority of PNH patients could benefit from greater access to therapy and the convenience of subcutaneous self-administration. We look forward to engaging with regulators around the world to advance into a registrational program for RA101495 in PNH. We believe the data announced today support the potential utility of RA101495 across the entire spectrum of C5-mediated diseases, including our clinical development programs in gMG, aHUS, and LN.”
|
Conference Call & Webcast |
||||
|
Date: |
Monday, December 4, 2017 | |||
|
Time: |
8:00 a.m. ET | |||
|
Telephone Access: |
Domestic callers, dial 844-419-1655, | |||
| International callers, dial 216-562-0467, | ||||
| Reference the Ra Pharmaceuticals conference call; | ||||
|
Telephone Access: |
Go to the Investor Relations section of the Ra Pharma website (http://ir.rapharma.com/phoenix.zhtml?c=254447&p=irol-calendar) and follow instructions for accessing the live webcast. Please connect to the website at least 15 minutes prior to the start of the conference call to ensure adequate time for any software download that may be necessary. |
|||
About RA101495
SC
Ra Pharma is developing RA101495 SC for paroxysmal
nocturnal hemoglobinuria (PNH), generalized
myasthenia gravis (gMG), atypical hemolytic uremic syndrome (aHUS),
and lupus
nephritis (LN). The product is designed for convenient, once-daily
subcutaneous self-administration. RA101495 SC is a synthetic,
macrocyclic peptide discovered using Ra Pharma’s powerful proprietary
drug discovery technology. The peptide binds complement component 5 (C5)
with sub-nanomolar affinity and allosterically inhibits its cleavage
into C5a and C5b upon activation of the classical, alternative, or
lectin pathways. By binding to a region of C5 corresponding to C5b,
RA101495 SC is designed to disrupt the interaction between C5b and C6
and prevent assembly of the membrane attack complex (MAC). This activity
may define an additional, novel mechanism for the inhibition of C5
function.
About RA101495 SC Phase 2 Clinical Program
The global,
dose-finding Phase 2 program is designed to evaluate the safety,
tolerability, preliminary efficacy, pharmacokinetics, and
pharmacodynamics of RA101495 SC in patients with PNH. The study is
evaluating RA101495 SC in three cohorts. Cohort A includes
eculizumab-naïve patients, Cohort B includes patients switching from
eculizumab to RA101495 SC, and a third cohort includes patients who are
currently treated with eculizumab, but have evidence of an inadequate
response. Patients in all three cohorts will be eligible for a long-term
extension study following the completion of the initial 12-week studies.
The primary efficacy endpoint is change in LDH from baseline to the mean
level from week 6 to week 12.
About Ra Pharmaceuticals
Ra Pharmaceuticals is a clinical
stage biopharmaceutical company focusing on the development of
next-generation therapeutics for complement-mediated diseases. The
Company discovers and develops peptides and small molecules to target
key components of the complement cascade.
Forward-Looking Statement
This press release contains
"forward-looking statements" within the meaning of the Private
Securities Litigation Reform Act of 1995, including, but not limited to,
statements regarding the safety, efficacy and regulatory and clinical
progress of our product candidates, including RA101495. All such
forward-looking statements are based on management's current
expectations of future events and are subject to a number of risks and
uncertainties that could cause actual results to differ materially and
adversely from those set forth in or implied by such forward-looking
statements. These risks and uncertainties include the risks that Ra
Pharma’s product candidates, including RA101495, will not successfully
be developed or commercialized; the risk that interim results as of
November 30, 2017 from the Company’s global Phase 2 clinical program
evaluating RA101495 for the treatment of PNH may not be indicative of
final study results; the risk that the interim results from the patient
enrollment and dosing to date may not be indicative of the data from the
full patient enrollment and dosing planned for such study; as well as
the other factors discussed in the “Risk Factors” section in Ra Pharma’s
most recently filed Annual Report on Form 10-K, as well as other risks
detailed in Ra Pharma’s subsequent filings with the Securities and
Exchange Commission. There can be no assurance that the actual results
or developments anticipated by Ra Pharma will be realized or, even if
substantially realized, that they will have the expected consequences
to, or effects on, Ra Pharma. All information in this press release is
as of the date of the release, and Ra Pharma undertakes no duty to
update this information unless required by law.