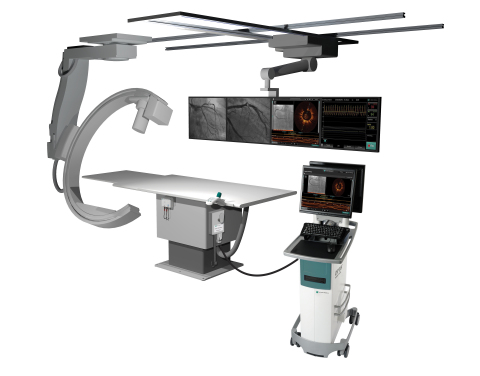
ST. PAUL, Minn.--(BUSINESS WIRE)--St. Jude Medical, Inc. (NYSE:STJ), a global medical device company, today announced the launch of the company’s OPTIS™ Mobile System in Japan and Europe. The diagnostic system is designed to couple state-of-the-art optical coherence tomography (OCT) and angiography co-registration with fractional flow reserve (FFR) technology into one portable system for hospitals with multiple catheterization labs.
Driven by a global increase in vascular disease and enabled by advances in technology, the catheterization lab allows physicians to use minimally invasive percutaneous coronary intervention (PCI) procedures that give patients an alternative to open heart surgery with the goal to improve patient outcomes, shorten hospital stays and reduce hospital costs. The latest OPTIS System offers physicians an efficient way to optimize PCI procedures for the treatment of vascular disease by combining diagnostic tools designed to improve patient outcomes into one portable device.
Utilizing a combination of technology previously available only in the St. Jude Medical OPTIS Integrated System, the OPTIS Mobile System is designed to help physicians make improved stenting decisions based on high-resolution and three dimensional OCT views of coronary anatomy while simultaneously mapping their exact location via angiogram. The OPTIS Mobile System also integrates St. Jude Medical PressureWire™ FFR measurement technology, which offers physicians detailed coronary hemodynamic (circulatory) information during PCI. Clinical data has shown the physiological measurements provided by St. Jude Medical FFR technology can improve outcomes and reduce health care costs over traditional diagnostic imaging tools.
“As the interventional cardiology landscape continues to expand, there is a real need for more portable intravascular imaging systems to ensure hospitals with multiple catheterization labs have the right technology available for physicians to make more informed treatment decisions during PCI,” said Dr. Nick West, Papworth Hospital, Cambridge, UK. “The imaging advancements offered with the OPTIS Mobile System provide the same benefits of the OPTIS Integrated System, and allow physicians to clearly visualize complex cardiac anatomy and evaluate how to best proceed during PCI.”
Sometimes referred to as coronary angioplasty, PCI is a catheter-based procedure designed to open coronary blockages or plaque buildup and restore blood flow to the heart. Traditionally, physicians have relied on either angiography or intravascular ultrasound to guide PCI procedures. St. Jude Medical’s ILUMIEN™ System first combined OCT and FFR technology to enable a more detailed, physiological and anatomical analysis of blood flow blockages inside the coronary vessels.
“The OPTIS Mobile System represents the ongoing commitment of St. Jude Medical to develop products that provide physicians with more options for improved OCT and angiography co-registration guidance in their daily practice,” said Dr. Eric Fain, Group President of St. Jude Medical. “In contrast with intravascular ultrasound, OCT offers increased resolution and highly-detailed intracoronary arterial views, which can improve PCI procedures, especially in complex cases.”
A System Backed by a Strong Research Portfolio
The benefits of utilizing OCT and FFR during PCI are supported by a robust portfolio of clinical data. The ILUMIEN I and ILUMIEN II studies have shown that St. Jude Medical’s OCT imaging technology can influence pre- and post-PCI decision making, stent sizing and deployment. The St. Jude Medical sponsored FAME body of evidence demonstrated St. Jude Medical PressureWire FFR measurement technology can improve patient outcomes and reduce costs in patients with stable coronary artery disease.
The multicenter ILUMIEN III: OPTIMIZE PCI clinical trial is currently underway to further develop the body of clinical evidence supporting OCT technology in relation to first generation imaging tools.
About Optical Coherence Tomography Imaging
St. Jude Medical’s OCT technology is an intravascular imaging tool that uses light to provide anatomical images of disease morphology and automated measurements. With OCT technology, physicians can visualize and measure important vessel characteristics that are otherwise not visible or difficult to assess with the older imaging technology. As a result, OCT can provide automated, highly accurate measurements that can help guide stent selection and deployment and assess stent placement to help ensure successful procedures. This can potentially minimize the need for repeat revascularization.
About Fractional Flow Reserve (FFR)
FFR is a physiological index used to determine the hemodynamic severity of narrowings (or lesions) in the coronary arteries, and is measured using St. Jude Medical PressureWire™ Aeris™ and PressureWire™ Certus™ systems. FFR specifically identifies which coronary narrowings are responsible for obstructing the flow of blood to a patient’s heart muscle (called ischemia), and helps guide the interventional cardiologist in determining which lesions warrant stenting, resulting in improved patient outcomes and reduced health care costs.
About St. Jude Medical
St. Jude Medical is a leading global medical device manufacturer and is dedicated to transforming the treatment of some of the world’s most expensive epidemic diseases. The company does this by developing cost-effective medical technologies that save and improve lives of patients around the world. Headquartered in St. Paul, Minn., St. Jude Medical has five major areas of focus that include heart failure, atrial fibrillation, neuromodulation, traditional cardiac rhythm management, and cardiovascular diseases. For more information, please visit sjm.com or follow us on Twitter @SJM_Media.
Forward-Looking Statements
This news release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 that involve risks and uncertainties. Such forward-looking statements include the expectations, plans and prospects for the Company, including potential clinical successes, anticipated regulatory approvals and future product launches, and projected revenues, margins, earnings and market shares. The statements made by the Company are based upon management’s current expectations and are subject to certain risks and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. These risks and uncertainties include market conditions and other factors beyond the Company’s control and the risk factors and other cautionary statements described in the Company’s filings with the SEC, including those described in the Risk Factors and Cautionary Statements sections of the Company’s Annual Report on Form 10-K for the fiscal year ended January 3, 2015 and Quarterly Report on Form 10-Q for the fiscal quarter ended October 3, 2015. The Company does not intend to update these statements and undertakes no duty to any person to provide any such update under any circumstance.